10.01.18
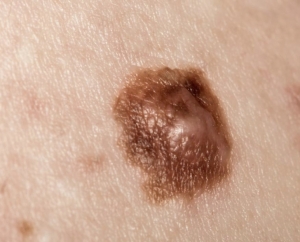
The US FDA has approved Libtayofor the treatment of patients with metastatic cutaneous squamous cell carcinoma (CSCC) or locally advanced CSCC who are not candidates for curative surgery or curative radiation.
CSCC is the second most common form of skin cancer and is responsible for an estimated 7,000 deaths each year in the US, and currently accounts for approximately 20 % of all skin cancers in the US. The number of newly diagnosed cases is expected to rise annually.
The FDA granted the approval of Libtayo to Regeneron Pharmaceuticals, Inc.
The drug is a fully-human monoclonal antibody targeting the immune checkpoint receptor PD-1 (programmed cell death protein-1) and is the first and only treatment specifically approved and available for advanced CSCC in the US. Libtayo works by targeting the cellular pathway known as PD-1 (protein found on the body’s immune cells and some cancer cells). By blocking this pathway, the drug may help the body’s immune system fight the cancer cells.
CSCC is the second most common form of skin cancer and is responsible for an estimated 7,000 deaths each year in the US, and currently accounts for approximately 20 % of all skin cancers in the US. The number of newly diagnosed cases is expected to rise annually.
The FDA granted the approval of Libtayo to Regeneron Pharmaceuticals, Inc.
The drug is a fully-human monoclonal antibody targeting the immune checkpoint receptor PD-1 (programmed cell death protein-1) and is the first and only treatment specifically approved and available for advanced CSCC in the US. Libtayo works by targeting the cellular pathway known as PD-1 (protein found on the body’s immune cells and some cancer cells). By blocking this pathway, the drug may help the body’s immune system fight the cancer cells.