Tom Schoenberg , Personal Care Consultant04.04.21
In today’s environment, producers of personal care products are continually challenged with new restrictions. The value of a product is no longer based on cost/performance, but whether it meets government safety requirements, which could be local, state or federal. Very often, the restrictions are not based on facts, but on potential risks.
For example, formaldehyde has been the most cost/effective preservative used in personal care for more than 80 years. Studies show that there is no evidence linking formaldehyde in cosmetics to cancer. Also, there is no official concentration at which formaldehyde is considered acceptable in personal care products. However, the name formaldehyde no longer appears on ingredient labels.1 The reason for this is that manufacturers prefer to avoid controversy. Once a chemical is deemed unsafe; e.g., formaldehyde, safety studies become irrelevant. Finding a replacement becomes the easier solution. Cost/performance becomes secondary.
Lately, several surface-active agents, (such as sulfates and nonionic), have been reported to be unsafe to the consumer. The following are three major surfactant classes in question:
• Lauryl Sulfates (sodium, ammonium, TEA, etc.): This class of compounds has been the workhorse in personal care cleansers for over 70 years. In a concentrated form, (greater than 1.0%), if left on the skin for a long time, (24 hours) sulfates will cause irritation.2 In the diluted form, such as, washing hair, face, or hands, the surfactant is soon rinsed off and there is no risk.3
Also, it has been rumored that “sulfates” are carcinogens but this myth is unfounded. Lauryl sulfates in a shampoo have no carcinogenic properties.3 At typical use levels, the Food and Drug Administration (FDA) and Cosmetic Ingredient Review Board (CIR) considered sulfates to be safe.
• Ethoxylates: Ethoxylated compounds (fatty alcohols, amines, etc.) are the other workhorse surfactants used in personal cleansers for over 60 years. 1,4 dioxane (a potential human carcinogen), a byproduct of the ethoxylation process, is present at trace levels in laureth sulfates. The surfactant industry has significantly reduced the level to less than 10ppm.4
The FDA conducted a skin absorption study which showed 1,4 dioxane can penetrate huma skin when applied in a lotion. However, further research determined that 1,4 dioxane evaporates readily, further diminishing the already small amount available for skin absorption, even in products that remain on skin for hours.
To date, there is no evidence that that 1,4 dioxane causes cancer in humans.4
• Alkanol Amides: The alkanol amides have been used for over 80 years in cleansing products. When the amide is combined with an anionic surfactant; e.g., lauryl sulfate, the combination produces higher viscosity and improved foam quality (height and stability). In the process of formulating amides, there is a chance to form a nitrosamine with the amide or free amine. Cocamide DEA is often the source of diethanolamine which forms nitrosamine-n-nitrosethanolamine (NDELA). The nitosating agent can come from various sources. One common source is a preservative, 2-bromo-2-nitropropane 1,3-diol.
Numerous studies link nitrosamines to cancer. Nitrosamines are listed as possible human carcinogens by the EPA, the International Agency for Research on Cancer, the US National Toxicity Program Report on Carcinogens and the California EPA Proposition 65 list of chemicals known to cause cancer or birth defects. Studies have also linked nitrosamines to developmental or reproductive toxicity, immunotoxicity, neurotoxicity and systemic toxicity.
The FDA began watching personal care products for nitrosamine in 1979, and published a report stating that products containing nitrosamine could be seen as adulterated and subject to FDA enforcement. This led to a striking drop of NDELA from 150ppm to 3ppm.5
An example of the formation of a nitrosamine is show in the following mechanism:
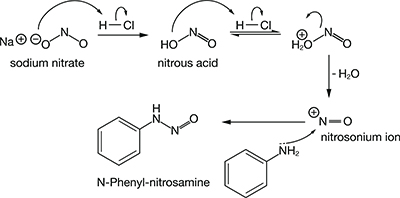
Mechanism of Nitrosamine Formation
Although there are other suspected toxic compounds found in personal care cleansers, the major ones of concern today are formaldehyde, sulfates, ethoxylates and nitrosamines. None have shown to be connected to human cancer at normal use levels for cleansing products. Industries response to this dilemma is to replace the suspected surfactant with an alternative. Alternative surfactants, such as isethionates, or sarcosinates, have resulted in higher cost and lower performance. One alternative that has not been fully investigated is Castile soap.6,7
Castile soap is a vegetable oil-based soap made in a style similar to that originating in the Castile region of Spain. Olive oil was the original vegetable oil used, however, eventually, olive oil was replaced with coconut oil. It is the most natural surfactant possible. The change in oils was most probably due to the better foaming properties of coconut oil soap, especially as coconut oil became easier to access.7
In the 1930s, in Springfield Massachusetts, Dr. John H. Breck founded Breck Shampoo Company. Breck shampoo was the first major promotion for a Castile Soap Shampoo in the US. Breck’s ad campaign, which began in the 1930s and continued through the 1980s, featured beautiful illustrated portraits of “Breck Girls,” who were meant to look like real life women instead of glamourous models. Later, in the 1940s they dismissed other leading shampoos as “basically, synthetic detergents” promising that their natural (Castile soap) formula would not over strip the hair.9 Castile soap shampoo eventually gave way to the synthetics (sulfates) in the 1970s when Breck switched from Castile to synthetic.
The Castile soap is a clear liquid around 40% active. It is prepared by the saponification of coconut or palm kernel oil (a triglyceride) with potassium hydroxide (note: sodium hydroxide will cause a cloudy product). The resulting compounds are potassium cocoate plus glycerin:
Coconut (or palm kernel) oil + KOH = K cocoate + glycerin
The glycerin is not removed. It provides a moisturizing effect to the skin.
When formulated, the coco sulfate is often combined with a foam booster and stabilizer which typically are amides or betaines. Cocamide DEA was the most common amide used, however, since DEA amides have been found to form nitrosamines, formulators have been searching to find alternatives. One replacement that appears promising, not based on DEA is: Cocamide DIPA (diisopropanolamide). It can often be used as a direct replacement for Cocamide DEA, without altering the performance of the product.
The following model formula is based on Castile soap which is the main cleansing agent and is lower cost compared to other alternatives. This formula is: sulfate free, ethoxylate free, 1,4-dioxane free, formaldehyde free, DEA free, nitrosamine free:
All-Purpose Cleaner
Procedure:
Combine the first two ingredients and water and blend. Heat to 40°C and blend until clear. Add preservative/fragrance and blend until clear. Cool and fill.
This formula can be used as a base for a variety of products: shampoos, hard surface cleaners, liquid hand soaps, etc. Also, other additives may be included: e.g., aloe, hemp oil, biocides, proteins, etc.
Personal cleansing manufacturers continue to face more restrictions on raw materials allowed in their products. Alternatives for sulfated fatty alcohols and alkanol amides are more expensive and have mediocre performance. Castile soap, a proven success for more than 80 years, has been found to be a low-cost replacement for sulfated alcohols. Also, Castile soap, the most natural surfactant available, is free of the claimed impurities, found in many personal care cleansers.
The past may be the solution.
References:
1. Lisette Mejia, “Formaldehyde in Cosmetics: What’s the Verdict?”, Women’s Health, Oct. 2011.
2. T.G. Schoenberg, “Sulfate-Free Cleansers”, Cosmetics and Toiletries, Dec. 2006, Vol 1`21, No. 12.
3. "The Truth About Sulfates”, Best Health, July 2019.
4. 1,4-Dioxane in Cosmetics: A Manufacturing Byproduct l EPA
5. Shelley R. Kramer, “Healthy – Communications.com, 2009
6. “How are Nitrosamines Formed? Bing
7. Schimmel Briefs, “Liquid Soaps”, April 1936
8. Wikipedia “Castile Soap”.
9. “How are nitrosamines formed?”, Bing
10. Breck, “It’s claim to fame: Breck Girls”
For example, formaldehyde has been the most cost/effective preservative used in personal care for more than 80 years. Studies show that there is no evidence linking formaldehyde in cosmetics to cancer. Also, there is no official concentration at which formaldehyde is considered acceptable in personal care products. However, the name formaldehyde no longer appears on ingredient labels.1 The reason for this is that manufacturers prefer to avoid controversy. Once a chemical is deemed unsafe; e.g., formaldehyde, safety studies become irrelevant. Finding a replacement becomes the easier solution. Cost/performance becomes secondary.
Surfactants
Lately, several surface-active agents, (such as sulfates and nonionic), have been reported to be unsafe to the consumer. The following are three major surfactant classes in question:
• Lauryl Sulfates (sodium, ammonium, TEA, etc.): This class of compounds has been the workhorse in personal care cleansers for over 70 years. In a concentrated form, (greater than 1.0%), if left on the skin for a long time, (24 hours) sulfates will cause irritation.2 In the diluted form, such as, washing hair, face, or hands, the surfactant is soon rinsed off and there is no risk.3
Also, it has been rumored that “sulfates” are carcinogens but this myth is unfounded. Lauryl sulfates in a shampoo have no carcinogenic properties.3 At typical use levels, the Food and Drug Administration (FDA) and Cosmetic Ingredient Review Board (CIR) considered sulfates to be safe.
• Ethoxylates: Ethoxylated compounds (fatty alcohols, amines, etc.) are the other workhorse surfactants used in personal cleansers for over 60 years. 1,4 dioxane (a potential human carcinogen), a byproduct of the ethoxylation process, is present at trace levels in laureth sulfates. The surfactant industry has significantly reduced the level to less than 10ppm.4
The FDA conducted a skin absorption study which showed 1,4 dioxane can penetrate huma skin when applied in a lotion. However, further research determined that 1,4 dioxane evaporates readily, further diminishing the already small amount available for skin absorption, even in products that remain on skin for hours.
To date, there is no evidence that that 1,4 dioxane causes cancer in humans.4
• Alkanol Amides: The alkanol amides have been used for over 80 years in cleansing products. When the amide is combined with an anionic surfactant; e.g., lauryl sulfate, the combination produces higher viscosity and improved foam quality (height and stability). In the process of formulating amides, there is a chance to form a nitrosamine with the amide or free amine. Cocamide DEA is often the source of diethanolamine which forms nitrosamine-n-nitrosethanolamine (NDELA). The nitosating agent can come from various sources. One common source is a preservative, 2-bromo-2-nitropropane 1,3-diol.
Numerous studies link nitrosamines to cancer. Nitrosamines are listed as possible human carcinogens by the EPA, the International Agency for Research on Cancer, the US National Toxicity Program Report on Carcinogens and the California EPA Proposition 65 list of chemicals known to cause cancer or birth defects. Studies have also linked nitrosamines to developmental or reproductive toxicity, immunotoxicity, neurotoxicity and systemic toxicity.
The FDA began watching personal care products for nitrosamine in 1979, and published a report stating that products containing nitrosamine could be seen as adulterated and subject to FDA enforcement. This led to a striking drop of NDELA from 150ppm to 3ppm.5
An example of the formation of a nitrosamine is show in the following mechanism:

Mechanism of Nitrosamine Formation
Castile soap is a vegetable oil-based soap made in a style similar to that originating in the Castile region of Spain. Olive oil was the original vegetable oil used, however, eventually, olive oil was replaced with coconut oil. It is the most natural surfactant possible. The change in oils was most probably due to the better foaming properties of coconut oil soap, especially as coconut oil became easier to access.7
In the 1930s, in Springfield Massachusetts, Dr. John H. Breck founded Breck Shampoo Company. Breck shampoo was the first major promotion for a Castile Soap Shampoo in the US. Breck’s ad campaign, which began in the 1930s and continued through the 1980s, featured beautiful illustrated portraits of “Breck Girls,” who were meant to look like real life women instead of glamourous models. Later, in the 1940s they dismissed other leading shampoos as “basically, synthetic detergents” promising that their natural (Castile soap) formula would not over strip the hair.9 Castile soap shampoo eventually gave way to the synthetics (sulfates) in the 1970s when Breck switched from Castile to synthetic.
The Castile soap is a clear liquid around 40% active. It is prepared by the saponification of coconut or palm kernel oil (a triglyceride) with potassium hydroxide (note: sodium hydroxide will cause a cloudy product). The resulting compounds are potassium cocoate plus glycerin:
Coconut (or palm kernel) oil + KOH = K cocoate + glycerin
The glycerin is not removed. It provides a moisturizing effect to the skin.
Other Additives
When formulated, the coco sulfate is often combined with a foam booster and stabilizer which typically are amides or betaines. Cocamide DEA was the most common amide used, however, since DEA amides have been found to form nitrosamines, formulators have been searching to find alternatives. One replacement that appears promising, not based on DEA is: Cocamide DIPA (diisopropanolamide). It can often be used as a direct replacement for Cocamide DEA, without altering the performance of the product.
The following model formula is based on Castile soap which is the main cleansing agent and is lower cost compared to other alternatives. This formula is: sulfate free, ethoxylate free, 1,4-dioxane free, formaldehyde free, DEA free, nitrosamine free:
All-Purpose Cleaner
| Ingredients | %Wt |
| Surfonate 40 (Potassium cocoate - 40%) (General Surfactants, Joliet IL) | 40.0 |
| Surfamide DIPA (Cocamide DIPA) (General Surfactants, Joliet IL) | 4.0 |
| Preservative/Fragrance q.s. | q.s. |
| Deionized water | q.s.to 100 |
Procedure:
Combine the first two ingredients and water and blend. Heat to 40°C and blend until clear. Add preservative/fragrance and blend until clear. Cool and fill.
This formula can be used as a base for a variety of products: shampoos, hard surface cleaners, liquid hand soaps, etc. Also, other additives may be included: e.g., aloe, hemp oil, biocides, proteins, etc.
Conclusion
Personal cleansing manufacturers continue to face more restrictions on raw materials allowed in their products. Alternatives for sulfated fatty alcohols and alkanol amides are more expensive and have mediocre performance. Castile soap, a proven success for more than 80 years, has been found to be a low-cost replacement for sulfated alcohols. Also, Castile soap, the most natural surfactant available, is free of the claimed impurities, found in many personal care cleansers.
The past may be the solution.
References:
1. Lisette Mejia, “Formaldehyde in Cosmetics: What’s the Verdict?”, Women’s Health, Oct. 2011.
2. T.G. Schoenberg, “Sulfate-Free Cleansers”, Cosmetics and Toiletries, Dec. 2006, Vol 1`21, No. 12.
3. "The Truth About Sulfates”, Best Health, July 2019.
4. 1,4-Dioxane in Cosmetics: A Manufacturing Byproduct l EPA
5. Shelley R. Kramer, “Healthy – Communications.com, 2009
6. “How are Nitrosamines Formed? Bing
7. Schimmel Briefs, “Liquid Soaps”, April 1936
8. Wikipedia “Castile Soap”.
9. “How are nitrosamines formed?”, Bing
10. Breck, “It’s claim to fame: Breck Girls”



















