Nava Dayan, Dr. Nava Dayan LLC11.02.15
Photo-toxicity reaction occurs when a biological tissue is exposed to a chemical entity which, when accompanied with radiation, becomes toxic. As such, only body organs that may be directly exposed to radiation, such as skin and eyes, are potential targets for photo-toxicity reaction. This does not mean that the chemical must be applied topically. In some cases it may be relevant to an oral or other systemic administration when a sufficient amount of the photo-toxic chemical accumulates in the skin from the blood. A compound will not be photo-toxic in the dark and in order to comply with the photo-toxicity definition it should undergo transformation to become harmful only when it interacts with radiation. A photo-toxic compound, therefore, should be photo-reactive and exposed to a radiation source; in nature, to sun light.
Photo-reactivity means that the compound undergoes modification that leads to biological toxicity. If it is not exposed to radiation; its toxicity will not be expressed. Some compounds can be both cytotoxic and photo-toxic. This means that they impart accelerated significant cell death to skin cells both in the dark and upon exposure to light. One example is Promethazine. Interestingly, one of the indications for promethazine is treatment of allergy symptoms such as itching, runny nose, sneezing, itchy or watery eyes, hives and itchy skin rashes. This may be perceived as a paradox because while this drug is aimed at alleviating skin inflammatory symptoms; under exposure to light it may actually provoke skin reaction. When considering drug use, in some cases, the benefit outweighs the adverse reaction tolerability; therefore an individual can decide if the risk of adverse reaction is acceptable since the relief of symptoms is profound and helpful.
Cosmetics are considered a luxury; therefore, the tolerability of skin adverse reactions should be reviewed in a different context and the risk associated with applying cosmetics should be reduced to a minimum. Furthermore, since administration of medication is taken under medical supervision and is accompanied with detailed instructions including potential adverse reactions anticipated; when used, the consumer is at least partially protected. In contrast, the use of cosmetic products is not monitored by a professional and misuse or abuse can rarely be predicted or controlled. If a cosmetic lotion contains a photo-toxic compound and the consumer applies it prior to using a tanning bed, the consequences can be harmful and the manufacturer does not have a layer of “protecting instructions” to prevent such potential scenario.
Sunscreen Paradox
Another paradox is that some sunscreens have been shown to be photo-toxic. While sunscreens are aimed to protect the skin from radiation, their ability to interact with light is the core of their activity and such can also be the core mechanism for a potential toxic effect. An example is the sunscreen ingredient Para-aminobenzoic acid that was shown in 1978 by Albert Kligman to exhibit photo-toxicity. In his study, Kligman found that amyl paradimethylaminobenzoate (Padimate A) was capable of provoking photo-toxic reaction. Following ultraviolet A (UVA) radiation at dosages of about 30 joules/cm2, a reaction grossly resembling sunburn developed in most subjects exposed to this sunscreen. The dominant histological feature was vacuolar degeneration of the epidermis without sunburn cells. Sunscreen products containing Padimate A were also shown to produce a similar reaction.
Kligman concluded that the product was ineffective. The similarity between sunburn and a photo-toxic response marks such products as harmful to be used upon sun exposure and not protective. For the record, formulators rarely use Padimate A. Photo-toxicity reactions can be classified into three types; the most common is photo-irritation but photo-sensitization and photo-genotoxicity are also possible. Photo-irritation is a light-induced, non-immunologic skin response to a photo-reactive compound. Photo-allergy is an acquired, immunologically mediated reaction to a chemical activated by light to form the allergenic compound (hapten). Mortality from photo-toxicity is rare and has been reported for example in individuals who were exposed to large doses of sunlight after taking the photo-toxic compound psoralene. A photo-toxic compound can become genotoxic and even carcinogenic when the exposure is prolonged and chronic.
For unknown reasons, men are found to have a higher probability than women to express a photo-allergic reaction. Age does not seem to play a role and drug induced photo-sensitivity can occur at any age. The clinical manifestation of a photo-irritation reaction resembles a moderate to severe sun burn. This reaction is clinically manifested as:
The deposition of a photo-toxic compound can be via application to skin and accumulation in the epidermis, but as mentioned previously, it can also occur if the photo-toxic compound is given orally. For example, oral tetracycline antibiotics administered for a sufficient time can result in a skin photo-toxic reaction due to its accumulation in the skin. Other compounds that were reported to be photo-toxic are selected essential oils, psoralens, sulfamides, coal tar derivatives and fluoroquinolones, as well as particular sunscreens such p-aminobenzoic acid, cinnamates and benzophenones.
Assessing Photo-toxicity
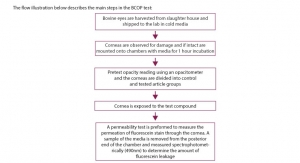
To determine if a compound carries the potential to be photo-toxic, a quantitative method is proposed by the OECD (OECD TG 101). This method utilizes a double-beam spectrophotometer which records the absorption differences between a blank and test solutions of the substance of interest to draw its spectrum. The guidelines describe determination of the ultraviolet-visible (UV-VIS) absorption spectrum of a chemical compound with the aim of providing an indication of the wavelengths at which the compound may be susceptible to a photochemical conversion. The test solutions should be prepared in a concentration which will result in at least one absorbance maximum in the range 0.5 to 1.5 units. If the tested article is not absorbing upon such analysis; it most likely does not carry a photo-toxic potential and further studies are required if the evaluating toxicologist decides to follow with additional assays. If significant absorption is incurred; additional studies to rule out photo-toxicity potential are required. The flow chart above describes the path for determination of photo-toxicity potential.
The in vitro study protocol OECD TG 432 that utilizes 3T3 mouse derived fibroblasts obtained regulatory acceptance in 2004. A photo-toxicity reaction is defined in the guidelines as “a toxic response from a substance applied to the body which is either elicited or increased (apparent at lower dose levels) after subsequent exposure to light or that is induced by skin irritation after systemic administration of a substance.”
The study is based on the detection of viable vs. dead cells utilizing 3T3 fibroblasts neutral red uptake. The neutral red (NR) uptake assay provides a quantitative estimation of the number of viable cells in a culture. It is based on the ability of viable cells to integrate the dye NR in the cellular lysosomes. The flow chat above describes the process. In brief, cells are seeded in tissue culture plates and are treated for the appropriate duration. The plates are then incubated with a medium containing NR. The cells are subsequently washed, the dye is extracted in each well and the absorbance is read using a spectrophotometer.
The assessment of photo-toxicity potential is brought up occasionally when companies are interested in skin care applications of edible compositions. It is often mistakenly thought that if a compound is non- harmful when ingested; it is ultimately non-harmful to be applied to skin. Photo-toxicity is one of the key endpoints for assessment when studying edible chemistries to be applied to skin. The assessment tools are relatively simple and can be tailored to an in vitro study tier.
References:
Nava Dayan
Owner
Dr. Nava Dayan LLC
Nava Dayan Ph.D. is the owner of Dr. Nava Dayan L.L.C, a skin science and research consultancy and serving the pharmaceutical, cosmetic, and personal care industries. She has 25 years of experience in the skin care segment, and more than 150 publication credits.
Tel: 201-206-7341
E-mail: nava.dayan@verizon.net
Photo-reactivity means that the compound undergoes modification that leads to biological toxicity. If it is not exposed to radiation; its toxicity will not be expressed. Some compounds can be both cytotoxic and photo-toxic. This means that they impart accelerated significant cell death to skin cells both in the dark and upon exposure to light. One example is Promethazine. Interestingly, one of the indications for promethazine is treatment of allergy symptoms such as itching, runny nose, sneezing, itchy or watery eyes, hives and itchy skin rashes. This may be perceived as a paradox because while this drug is aimed at alleviating skin inflammatory symptoms; under exposure to light it may actually provoke skin reaction. When considering drug use, in some cases, the benefit outweighs the adverse reaction tolerability; therefore an individual can decide if the risk of adverse reaction is acceptable since the relief of symptoms is profound and helpful.
Cosmetics are considered a luxury; therefore, the tolerability of skin adverse reactions should be reviewed in a different context and the risk associated with applying cosmetics should be reduced to a minimum. Furthermore, since administration of medication is taken under medical supervision and is accompanied with detailed instructions including potential adverse reactions anticipated; when used, the consumer is at least partially protected. In contrast, the use of cosmetic products is not monitored by a professional and misuse or abuse can rarely be predicted or controlled. If a cosmetic lotion contains a photo-toxic compound and the consumer applies it prior to using a tanning bed, the consequences can be harmful and the manufacturer does not have a layer of “protecting instructions” to prevent such potential scenario.
Sunscreen Paradox
Another paradox is that some sunscreens have been shown to be photo-toxic. While sunscreens are aimed to protect the skin from radiation, their ability to interact with light is the core of their activity and such can also be the core mechanism for a potential toxic effect. An example is the sunscreen ingredient Para-aminobenzoic acid that was shown in 1978 by Albert Kligman to exhibit photo-toxicity. In his study, Kligman found that amyl paradimethylaminobenzoate (Padimate A) was capable of provoking photo-toxic reaction. Following ultraviolet A (UVA) radiation at dosages of about 30 joules/cm2, a reaction grossly resembling sunburn developed in most subjects exposed to this sunscreen. The dominant histological feature was vacuolar degeneration of the epidermis without sunburn cells. Sunscreen products containing Padimate A were also shown to produce a similar reaction.
Kligman concluded that the product was ineffective. The similarity between sunburn and a photo-toxic response marks such products as harmful to be used upon sun exposure and not protective. For the record, formulators rarely use Padimate A. Photo-toxicity reactions can be classified into three types; the most common is photo-irritation but photo-sensitization and photo-genotoxicity are also possible. Photo-irritation is a light-induced, non-immunologic skin response to a photo-reactive compound. Photo-allergy is an acquired, immunologically mediated reaction to a chemical activated by light to form the allergenic compound (hapten). Mortality from photo-toxicity is rare and has been reported for example in individuals who were exposed to large doses of sunlight after taking the photo-toxic compound psoralene. A photo-toxic compound can become genotoxic and even carcinogenic when the exposure is prolonged and chronic.
For unknown reasons, men are found to have a higher probability than women to express a photo-allergic reaction. Age does not seem to play a role and drug induced photo-sensitivity can occur at any age. The clinical manifestation of a photo-irritation reaction resembles a moderate to severe sun burn. This reaction is clinically manifested as:
- Redness;
- Itching;
- Vesicular lesions which become scaly, crusted or hardened;
- For photo-allergic reaction an induction phase before elicitation is required.
The deposition of a photo-toxic compound can be via application to skin and accumulation in the epidermis, but as mentioned previously, it can also occur if the photo-toxic compound is given orally. For example, oral tetracycline antibiotics administered for a sufficient time can result in a skin photo-toxic reaction due to its accumulation in the skin. Other compounds that were reported to be photo-toxic are selected essential oils, psoralens, sulfamides, coal tar derivatives and fluoroquinolones, as well as particular sunscreens such p-aminobenzoic acid, cinnamates and benzophenones.
Assessing Photo-toxicity
To determine if a compound carries the potential to be photo-toxic, a quantitative method is proposed by the OECD (OECD TG 101). This method utilizes a double-beam spectrophotometer which records the absorption differences between a blank and test solutions of the substance of interest to draw its spectrum. The guidelines describe determination of the ultraviolet-visible (UV-VIS) absorption spectrum of a chemical compound with the aim of providing an indication of the wavelengths at which the compound may be susceptible to a photochemical conversion. The test solutions should be prepared in a concentration which will result in at least one absorbance maximum in the range 0.5 to 1.5 units. If the tested article is not absorbing upon such analysis; it most likely does not carry a photo-toxic potential and further studies are required if the evaluating toxicologist decides to follow with additional assays. If significant absorption is incurred; additional studies to rule out photo-toxicity potential are required. The flow chart above describes the path for determination of photo-toxicity potential.
The in vitro study protocol OECD TG 432 that utilizes 3T3 mouse derived fibroblasts obtained regulatory acceptance in 2004. A photo-toxicity reaction is defined in the guidelines as “a toxic response from a substance applied to the body which is either elicited or increased (apparent at lower dose levels) after subsequent exposure to light or that is induced by skin irritation after systemic administration of a substance.”
The study is based on the detection of viable vs. dead cells utilizing 3T3 fibroblasts neutral red uptake. The neutral red (NR) uptake assay provides a quantitative estimation of the number of viable cells in a culture. It is based on the ability of viable cells to integrate the dye NR in the cellular lysosomes. The flow chat above describes the process. In brief, cells are seeded in tissue culture plates and are treated for the appropriate duration. The plates are then incubated with a medium containing NR. The cells are subsequently washed, the dye is extracted in each well and the absorbance is read using a spectrophotometer.
The assessment of photo-toxicity potential is brought up occasionally when companies are interested in skin care applications of edible compositions. It is often mistakenly thought that if a compound is non- harmful when ingested; it is ultimately non-harmful to be applied to skin. Photo-toxicity is one of the key endpoints for assessment when studying edible chemistries to be applied to skin. The assessment tools are relatively simple and can be tailored to an in vitro study tier.
References:
- OECD TG 432
- OECD TG 101
- Kaidbey KH, Kligman AM. Phototoxicity to a sunscreen ingredient. Padimate A. Arch Dermatol. 1978 Apr;114(4):547-9.
Nava Dayan
Owner
Dr. Nava Dayan LLC
Nava Dayan Ph.D. is the owner of Dr. Nava Dayan L.L.C, a skin science and research consultancy and serving the pharmaceutical, cosmetic, and personal care industries. She has 25 years of experience in the skin care segment, and more than 150 publication credits.
Tel: 201-206-7341
E-mail: nava.dayan@verizon.net