Nava Dayan, Dr. Nava Dayan LLC01.04.16
Skin sensitization (allergy) is one of the most problematic endpoints to evaluate in the safety assessment of skin exposure. It is a very complex reaction that often involves the humoral (blood) and lymphatic (immune) systems. It can be unpredictable and may present cross reactivity with other organs such as lung or digestive system. It is perhaps the only cosmetic adverse reaction that may lead rapidly to mortality. Although rare, an anaphylaxis to skin care products is certainly possible. The fact that Western population presents steady growth in expressing sensitization adds to the importance of such assessment. The Japanese National Institute of Health Sciences reported in 2014 that in 2009 a case in which two patients who had been primary sensitized to hydrolyzed wheat protein (HWP) present in facial soap subsequently experienced wheat-dependent exercise-induced anaphylaxis (WDEIA) after the ingestion of wheat products. Since 2009, notes the Institute, more than 1,900 patients with such an allergy have been reported (through June 20, 2013) from various institutes all around Japan. Importantly, the majority of the patients used the same facial soap (Cha no Shizuku) containing acid-hydrolyzed wheat protein (acid-HWP). The commercial acid-HWP contained in the facial soap (Glupearl 19S) of average molecular weight of 30-50 kDa was produced from gluten.
Although an allergy from wheat may not be surprising, another interesting report in Japan points toward a case of anaphylaxis to a compound that may be perceived as non-harmful, polyethylene glycol analogue. A different paper from Turkey describes severe allergic reaction of an infant to cologne; the subject developed anaphylaxis after his mother applied cologne to his face. The infant was described as an otherwise healthy two-month old and was admitted to the emergency room with facial edema and shortness of breath. He was treated with a variety of drugs and was discharged after symptoms regressed.
Finally an interesting case of anaphylaxis to the preservative 2-phenoxyethanol has been described in Spain in 2010 in a 42 year-old woman who was exposed to a variety of cosmetic products applied to body and face. In one incident, when applying a moisturizer to her arms and legs, the woman developed immediate hives followed by dizziness. An open test on the volar forearm confirmed immediate positivity to the preservative phenoxyethanol.
Classifying Reactions
Skin allergy reaction can be broadly classified as immediate or delayed. The immediate reaction involves activation of mast cells that reside in the dermis. Such activation can be mediated via antibodies that are generated after at least one prior incidence of exposure or directly to the cell. A delayed reaction involves the activation of T-cells in the lymphatic system and is mediated by antibodies as well. The immediate sensitization reaction is also termed “type 1” or “urticaria” and the delayed reaction termed “type 4.” The clinical exposure test, Repeat Insult Patch Test (RIPT), which is used extensively to test ingredients and formulations, does not evaluate an immediate sensitization reaction, only the delayed type. Apart from basic research-developed assays by a few Academic setups or the clinical skin prick test which involves puncturing the skin with a needle or pin containing a small amount of the tested allergen, there are no commercial assays for type 1 reaction and cosmetic ingredients and formulations are not routinely evaluated for such.
A few prerequisites are considered in the evaluation of a potential allergenicity. To provoke such reaction, the allergen should penetrate the stratum corneum at sufficient levels and interact with carrier proteins in the skin. Therefore, the reaction will emerge only when exposure involves sufficient quantities combined with specificity of the individual.
An in vitro assay for the assessment of type 4 skin sensitization called ARE-Nrf2 Luciferase Test Method is based on the Keap1-Nrf2-antioxidant/electrophile response element (ARE) pathways. These pathways are involved in the cytoprotective response to oxidative stress and are also known to be involved in the cellular processes in skin sensitization. The assay uses the HaCaT immortalized human keratinocytes cell line transfected with a selective plasmid to quantify the induction of a luciferase gene as a measure of the ARE pathway. Keap-1 is a sensor protein that contains a highly reactive cysteine but not lysine. It targets the transcription factor Nrf2 (nuclear factor erythroid 2-related factor 2). Nrf2 is essential for the expression of full sensitization and a condition for the allergen induced INF-γ formation. Since sensitizers can be reactive to either lysine or cysteine in the skin protein reaction, it is important to note that potential sensitizers with exclusive reactivity toward lysine will not induce the Keap1 pathway and thus will not be identified by this assay. The mediators involved in the lysine path are yet to be explored.
OECD test guidelines number 442 D for ARE-Nrf2 Luciferase Test Method were issued on February 5, 2015. The assay is based on the findings that skin sensitizers may act on the sensor protein Keap 1 and, via covalent bonding, modify its structure resulting in dissociation from the transcription factor Nrf2. The dissociated Nrf2 can proceed in activation of ARE dependent genes. One of the advantages of this method is the ability to classify potential sensitizers on a scale from weak to strong. Examples for sensitizers identified by this method are: weak: cinnamyl alcohol and cinnamic aldehyde, moderate: 2-mercaptobenzothiazole and strong: methyl dibromo glutaronitrite.
Another relatively simple, quick and inexpensive assay to assess potential skin sensitization is the direct peptide reactivity assay (DPRA). This assay is based on the understanding that in order to provoke a cascade of immune response to generate antibodies; the sensitizing compound or the essential part in the molecule that is recognized as the sensitizer must bind to a protein carrier. This process is named haptenization and the sensitizing chemistry is termed hapten. DPRA is designed to mimic the covalent bonding of electrophilic chemicals (sensitizers) to nucleophilic domains present in skin proteins. It allows the quantification of the chemical reactivity toward model synthetic peptides containing cysteine and lysine. The assay involves incubation of the tested compounds with synthetic peptides containing either cysteine or lysine at single concentration in triplicates for 24 hours and the assessed endpoint is conducted by HPLC/UV to quantify the percent depletion of cysteine and lysine relative to the vehicle control. The DRPA was published on Feb. 5, 2015 as OECD 442C.
These two in vitro assays are part of a suggested battery of available studies. The studies are at different stages of evaluation and validation. Additional studies proposed are based on gene expression and cell activation as well as on cytokine release. Still missing in the proposed battery is the assessment of mast cells involvement in the reaction; key cell population in the dermis that mediates allergy and especially the type 1 immediate reaction. To date, there is no single assay that can predict all possible potential scenarios of skin sensitization and each assay has limitations that should be understood and considered, therefore in such assessment a battery of assays should be developed and conducted. The conducted study tier should be customized based on risk assessment of exposure and other attributes associated with the tested composition. Conducting in silico quantitative structure activity relationships (qSAR) is highly recommended to fine-tune assay selection.
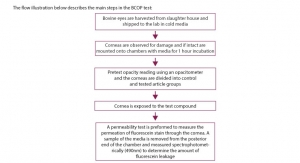
The author would like to thank Cyprotex Discovery Ltd (www.cyprotex.com) for providing some data as well as the image that appears in the column.
References
Kobayashi T et al. Int J Dermatol. 54(8): (2015) e302-5.
Yamasuji Y et al. Contact Dermatitis 69(3): (2013) 183-5.
Higashi Y et al Allergol Immunopathol 40(3) (2012) 198.
Arikan-Ayyildiz Z et al J Investig Allergol Clin Immunol.;21(6): (2011) 494-5.
Núñez Orjales R et al J Investig Allergol Clin Immunol.;20(4): (2010) 354-355.
http://www.oecd-ilibrary.org/environment/test-no-442d-in-vitro-skin-sensitisation_9789264229822-en
http://www.oecd.org/env/test-no-442c-in-chemico-skin-sensitisation-9789264229709-en.htm
Natsch A. Tox Sci. 113(2) (2010) 284-292.
Nava Dayan
Owner
Dr. Nava Dayan LLC
Nava Dayan Ph.D. is the owner of Dr. Nava Dayan L.L.C, a skin science and research consultancy and serving the pharmaceutical, cosmetic, and personal care industries. She has 25 years of experience in the skin care segment, and more than 150 publication credits.
Tel: 201-206-7341
E-mail: nava.dayan@verizon.net
Although an allergy from wheat may not be surprising, another interesting report in Japan points toward a case of anaphylaxis to a compound that may be perceived as non-harmful, polyethylene glycol analogue. A different paper from Turkey describes severe allergic reaction of an infant to cologne; the subject developed anaphylaxis after his mother applied cologne to his face. The infant was described as an otherwise healthy two-month old and was admitted to the emergency room with facial edema and shortness of breath. He was treated with a variety of drugs and was discharged after symptoms regressed.
Finally an interesting case of anaphylaxis to the preservative 2-phenoxyethanol has been described in Spain in 2010 in a 42 year-old woman who was exposed to a variety of cosmetic products applied to body and face. In one incident, when applying a moisturizer to her arms and legs, the woman developed immediate hives followed by dizziness. An open test on the volar forearm confirmed immediate positivity to the preservative phenoxyethanol.
Classifying Reactions
Skin allergy reaction can be broadly classified as immediate or delayed. The immediate reaction involves activation of mast cells that reside in the dermis. Such activation can be mediated via antibodies that are generated after at least one prior incidence of exposure or directly to the cell. A delayed reaction involves the activation of T-cells in the lymphatic system and is mediated by antibodies as well. The immediate sensitization reaction is also termed “type 1” or “urticaria” and the delayed reaction termed “type 4.” The clinical exposure test, Repeat Insult Patch Test (RIPT), which is used extensively to test ingredients and formulations, does not evaluate an immediate sensitization reaction, only the delayed type. Apart from basic research-developed assays by a few Academic setups or the clinical skin prick test which involves puncturing the skin with a needle or pin containing a small amount of the tested allergen, there are no commercial assays for type 1 reaction and cosmetic ingredients and formulations are not routinely evaluated for such.
A few prerequisites are considered in the evaluation of a potential allergenicity. To provoke such reaction, the allergen should penetrate the stratum corneum at sufficient levels and interact with carrier proteins in the skin. Therefore, the reaction will emerge only when exposure involves sufficient quantities combined with specificity of the individual.
An in vitro assay for the assessment of type 4 skin sensitization called ARE-Nrf2 Luciferase Test Method is based on the Keap1-Nrf2-antioxidant/electrophile response element (ARE) pathways. These pathways are involved in the cytoprotective response to oxidative stress and are also known to be involved in the cellular processes in skin sensitization. The assay uses the HaCaT immortalized human keratinocytes cell line transfected with a selective plasmid to quantify the induction of a luciferase gene as a measure of the ARE pathway. Keap-1 is a sensor protein that contains a highly reactive cysteine but not lysine. It targets the transcription factor Nrf2 (nuclear factor erythroid 2-related factor 2). Nrf2 is essential for the expression of full sensitization and a condition for the allergen induced INF-γ formation. Since sensitizers can be reactive to either lysine or cysteine in the skin protein reaction, it is important to note that potential sensitizers with exclusive reactivity toward lysine will not induce the Keap1 pathway and thus will not be identified by this assay. The mediators involved in the lysine path are yet to be explored.
OECD test guidelines number 442 D for ARE-Nrf2 Luciferase Test Method were issued on February 5, 2015. The assay is based on the findings that skin sensitizers may act on the sensor protein Keap 1 and, via covalent bonding, modify its structure resulting in dissociation from the transcription factor Nrf2. The dissociated Nrf2 can proceed in activation of ARE dependent genes. One of the advantages of this method is the ability to classify potential sensitizers on a scale from weak to strong. Examples for sensitizers identified by this method are: weak: cinnamyl alcohol and cinnamic aldehyde, moderate: 2-mercaptobenzothiazole and strong: methyl dibromo glutaronitrite.
Another relatively simple, quick and inexpensive assay to assess potential skin sensitization is the direct peptide reactivity assay (DPRA). This assay is based on the understanding that in order to provoke a cascade of immune response to generate antibodies; the sensitizing compound or the essential part in the molecule that is recognized as the sensitizer must bind to a protein carrier. This process is named haptenization and the sensitizing chemistry is termed hapten. DPRA is designed to mimic the covalent bonding of electrophilic chemicals (sensitizers) to nucleophilic domains present in skin proteins. It allows the quantification of the chemical reactivity toward model synthetic peptides containing cysteine and lysine. The assay involves incubation of the tested compounds with synthetic peptides containing either cysteine or lysine at single concentration in triplicates for 24 hours and the assessed endpoint is conducted by HPLC/UV to quantify the percent depletion of cysteine and lysine relative to the vehicle control. The DRPA was published on Feb. 5, 2015 as OECD 442C.
These two in vitro assays are part of a suggested battery of available studies. The studies are at different stages of evaluation and validation. Additional studies proposed are based on gene expression and cell activation as well as on cytokine release. Still missing in the proposed battery is the assessment of mast cells involvement in the reaction; key cell population in the dermis that mediates allergy and especially the type 1 immediate reaction. To date, there is no single assay that can predict all possible potential scenarios of skin sensitization and each assay has limitations that should be understood and considered, therefore in such assessment a battery of assays should be developed and conducted. The conducted study tier should be customized based on risk assessment of exposure and other attributes associated with the tested composition. Conducting in silico quantitative structure activity relationships (qSAR) is highly recommended to fine-tune assay selection.
The author would like to thank Cyprotex Discovery Ltd (www.cyprotex.com) for providing some data as well as the image that appears in the column.
References
Kobayashi T et al. Int J Dermatol. 54(8): (2015) e302-5.
Yamasuji Y et al. Contact Dermatitis 69(3): (2013) 183-5.
Higashi Y et al Allergol Immunopathol 40(3) (2012) 198.
Arikan-Ayyildiz Z et al J Investig Allergol Clin Immunol.;21(6): (2011) 494-5.
Núñez Orjales R et al J Investig Allergol Clin Immunol.;20(4): (2010) 354-355.
http://www.oecd-ilibrary.org/environment/test-no-442d-in-vitro-skin-sensitisation_9789264229822-en
http://www.oecd.org/env/test-no-442c-in-chemico-skin-sensitisation-9789264229709-en.htm
Natsch A. Tox Sci. 113(2) (2010) 284-292.
Nava Dayan
Owner
Dr. Nava Dayan LLC
Nava Dayan Ph.D. is the owner of Dr. Nava Dayan L.L.C, a skin science and research consultancy and serving the pharmaceutical, cosmetic, and personal care industries. She has 25 years of experience in the skin care segment, and more than 150 publication credits.
Tel: 201-206-7341
E-mail: nava.dayan@verizon.net