Nadim Shaath, Alpha Research & Development Ltd06.01.18
Citing serious, long-term environmental damage to coral reefs and to the oceans, Hawaii lawmakers passed a bill in May that bans the use of two ultraviolet filters, oxybenzone and octinoxate. If this bill is signed by Hawaii’s governor, the ban will become effective on January 1, 2021.1
Take a deep breath, and let it out slowly. If, and when, the ban extends to Florida and California, this may signal the beginning of the demise of the sunscreen industry.
Why? Simply stated, those two ingredients are in roughly 65% of sunscreens sold today. Their removal will necessitate the reformulation of all the top-selling sunscreen products on the market. Take as an example the Helioplex patent, which is the cornerstone of Neutrogena’s sun care products. It would be rendered useless as oxybenzone is crucial in this patented formulation. So is octinoxate. Also, the ever-popular spray sunscreen products would have to be reformulated. That will be a formidable task as we do not have effective UV filters currently approved by the FDA. All we would be left with are primarily the inorganic particulates of zinc oxide and titanium oxides.
Some of you may be asking, “Weren’t there 21 UV filters approved as Category I by the FDA? Didn’t the FDA graciously add two more ingredients, namely avobenzone and zinc oxide, in the 1990s? Aren’t there eight new UV filters awaiting approval by the FDA under the so-called Time and Extent Application (TEA)? Aren’t the laboratories in the US buzzing with research and activity to introduce new and improved UV filters?”
The truth of the matter is that only five ingredients—I repeat, FIVE—out of the 23 originally approved Category I ingredients are potential UV filters for future sunscreen products. The eight TEA ingredients have been recently rejected by the FDA, which will require additional massive amounts of information as a requirement for future approval. Finally, there are no ingredients—I repeat, NO INGREDIENTS—on the horizon that are emanating from US research laboratories! It is so sad that it is beyond description.
At the risk of repeating myself after writing column after column the last dozen years delineating the dilemma the sunscreen industry is in, I will walk you through it one more time.
The Precarious Status of Category I UV Filters
The majority of those 23 UV filters are based on the chemistries from the 1950s and 1960s. They are archaic, ineffective and, in many cases, unsafe or unstable. Many of those ingredients have given the sunscreen industry a blackeye. Most are small molecules that tend to penetrate the skin, some are photo-unstable unless quenched with other UV filters, and a few others, including oxybenzone, have been demonstrated to be endocrine disruptors. With the exception of zinc oxide and avobenzone, the remaining list of filters protects only against the UVB rays; oxybenzone and menthyl anthranilate exhibit weak UVA protection.
UVA protection is acknowledged by most experts as crucial and pivotal in skin cancer protection. We simply do not have adequate UVA protection in the US. Avobenzone is photo-unstable and unless it is propped up with UV quenchers, it’s totally useless. Zinc oxide has some UVA protection, but formulating with it is tedious and tends to conglomerate, rendering it ineffective over time. Of course, zinc oxide—unless micronized effectively—will impart cosmetically unappealing opaqueness to the skin upon application. Micronization helps, but nanoparticles pose potential hazards, the extent of which has not been yet determined.
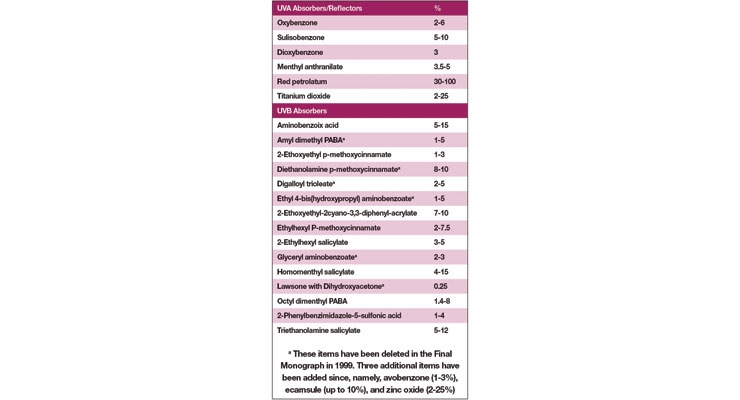
Table I lists the 21 category I ingredients originally approved by the FDA.2 You will note immediately that the only ingredients remaining that are potential UV filters are octyl salicylate, homomenthyl salicylate and titanium dioxide. Add to this list the more recently approved avobenzone and zinc oxide. These are the five ingredients that are possible to use today for all sun care products. An argument could be made to use other ingredients on Table I that have gone by the wayside in the last dozen years such as octyl dimethyl PABA or menthyl anthranilate, but it is certainly a stretch.
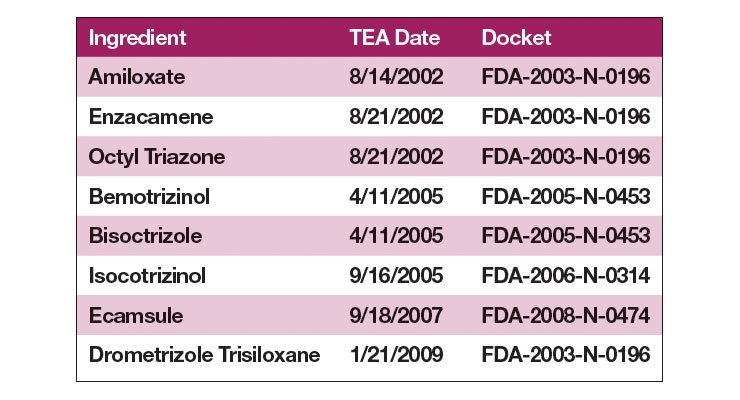
Table II lists the eight TEA ingredients that have enjoyed popularity in Europe and in the rest of the world, but the FDA deemed recently that they may not be used in the US today.3 Several of those ingredients are excellent UVA ingredients that have been designed based on the 500 Dalton rule that ensures that they do not penetrate the skin. They have a worldwide history of use exceeding a dozen years or more. The FDA, however, views all sunscreen filters as drugs, so their approval process is subject to the same rigorous standards as a diabetes or heart drug, a cholesterol drug, or even a cancer drug.
The bar is currently held much too high for sunscreens. If designed properly—unlike most of the currently approved UV filters in the US—sunscreens would not require such rigorous testing if they are truly safe, high molecular weight filters that do not penetrate the skin upon application. Those ingredients, in my opinion, under the present dire circumstances should be approved under the current cosmetics standards and not as drugs. Within a relatively short time period, we would once again have a healthy list of UVA and UVB filters that would allow for the immediate reformulation of our currently archaic sunscreens. These products would be safe, stable, broad spectrum and effective in reducing the spiraling epidemic of skin cancer.
References:
Nadim Shaath
Alpha Research & Development Ltd
Email: alpharnd@aol.com
Dr. Nadim Shaath is the president of Alpha Research & Development, Ltd. in White Plains, NY. He has over 30 years of experience as chairman of the chemistry department at SUNY-Purchase and the CEO of Kato Worldwide. Recently he published his new book entitled “Healing Civilizations: The Search for Therapeutic Essential Oils and Nutrients” Cameron Books, Petaluma, CA.
Take a deep breath, and let it out slowly. If, and when, the ban extends to Florida and California, this may signal the beginning of the demise of the sunscreen industry.
Why? Simply stated, those two ingredients are in roughly 65% of sunscreens sold today. Their removal will necessitate the reformulation of all the top-selling sunscreen products on the market. Take as an example the Helioplex patent, which is the cornerstone of Neutrogena’s sun care products. It would be rendered useless as oxybenzone is crucial in this patented formulation. So is octinoxate. Also, the ever-popular spray sunscreen products would have to be reformulated. That will be a formidable task as we do not have effective UV filters currently approved by the FDA. All we would be left with are primarily the inorganic particulates of zinc oxide and titanium oxides.
Some of you may be asking, “Weren’t there 21 UV filters approved as Category I by the FDA? Didn’t the FDA graciously add two more ingredients, namely avobenzone and zinc oxide, in the 1990s? Aren’t there eight new UV filters awaiting approval by the FDA under the so-called Time and Extent Application (TEA)? Aren’t the laboratories in the US buzzing with research and activity to introduce new and improved UV filters?”
The truth of the matter is that only five ingredients—I repeat, FIVE—out of the 23 originally approved Category I ingredients are potential UV filters for future sunscreen products. The eight TEA ingredients have been recently rejected by the FDA, which will require additional massive amounts of information as a requirement for future approval. Finally, there are no ingredients—I repeat, NO INGREDIENTS—on the horizon that are emanating from US research laboratories! It is so sad that it is beyond description.
At the risk of repeating myself after writing column after column the last dozen years delineating the dilemma the sunscreen industry is in, I will walk you through it one more time.
The Precarious Status of Category I UV Filters
The majority of those 23 UV filters are based on the chemistries from the 1950s and 1960s. They are archaic, ineffective and, in many cases, unsafe or unstable. Many of those ingredients have given the sunscreen industry a blackeye. Most are small molecules that tend to penetrate the skin, some are photo-unstable unless quenched with other UV filters, and a few others, including oxybenzone, have been demonstrated to be endocrine disruptors. With the exception of zinc oxide and avobenzone, the remaining list of filters protects only against the UVB rays; oxybenzone and menthyl anthranilate exhibit weak UVA protection.
UVA protection is acknowledged by most experts as crucial and pivotal in skin cancer protection. We simply do not have adequate UVA protection in the US. Avobenzone is photo-unstable and unless it is propped up with UV quenchers, it’s totally useless. Zinc oxide has some UVA protection, but formulating with it is tedious and tends to conglomerate, rendering it ineffective over time. Of course, zinc oxide—unless micronized effectively—will impart cosmetically unappealing opaqueness to the skin upon application. Micronization helps, but nanoparticles pose potential hazards, the extent of which has not been yet determined.
Table I lists the 21 category I ingredients originally approved by the FDA.2 You will note immediately that the only ingredients remaining that are potential UV filters are octyl salicylate, homomenthyl salicylate and titanium dioxide. Add to this list the more recently approved avobenzone and zinc oxide. These are the five ingredients that are possible to use today for all sun care products. An argument could be made to use other ingredients on Table I that have gone by the wayside in the last dozen years such as octyl dimethyl PABA or menthyl anthranilate, but it is certainly a stretch.
Table II lists the eight TEA ingredients that have enjoyed popularity in Europe and in the rest of the world, but the FDA deemed recently that they may not be used in the US today.3 Several of those ingredients are excellent UVA ingredients that have been designed based on the 500 Dalton rule that ensures that they do not penetrate the skin. They have a worldwide history of use exceeding a dozen years or more. The FDA, however, views all sunscreen filters as drugs, so their approval process is subject to the same rigorous standards as a diabetes or heart drug, a cholesterol drug, or even a cancer drug.
The bar is currently held much too high for sunscreens. If designed properly—unlike most of the currently approved UV filters in the US—sunscreens would not require such rigorous testing if they are truly safe, high molecular weight filters that do not penetrate the skin upon application. Those ingredients, in my opinion, under the present dire circumstances should be approved under the current cosmetics standards and not as drugs. Within a relatively short time period, we would once again have a healthy list of UVA and UVB filters that would allow for the immediate reformulation of our currently archaic sunscreens. These products would be safe, stable, broad spectrum and effective in reducing the spiraling epidemic of skin cancer.
References:
- https://www.chemistryworld.com.
- Shaath, Nadim A., The Chemistry of Ultraviolet Filters, Principles and Practice of Photoprotection Springer International Publishing, pp 143-157 (2016)
- Shaath, Nadim A., The Sunscreen Filter, Happi Magazine, October 2014.
Nadim Shaath
Alpha Research & Development Ltd
Email: alpharnd@aol.com
Dr. Nadim Shaath is the president of Alpha Research & Development, Ltd. in White Plains, NY. He has over 30 years of experience as chairman of the chemistry department at SUNY-Purchase and the CEO of Kato Worldwide. Recently he published his new book entitled “Healing Civilizations: The Search for Therapeutic Essential Oils and Nutrients” Cameron Books, Petaluma, CA.