Science Programs at University of Cincinnati, James L. Winkle College of Pharmacy • Deepika Raina, L’Oreal • Geoff Waby, Advanced Testing Laboratory • Michael Loewenstein, Q Laboratories LLC • Ly Tran-Osowski, Troy-an Arxada Company
Manufacturing Hygiene Task Force • PCPC Microbiology & Quality Committees
This is the third and final part of a series describing the concept of the manufacturing hygienist. As detailed in Parts I and II of this series,1,2 we liken the manufacturing hygienist to a conductor of an orchestra— organizing the disparate elements of both product development and manufacturing, to drive harmonious systems and deliver high quality products to the consumer. Here, we detail the risk against which the manufacturing hygienist labors and introduce the necessary training and credentialing certification programs that qualify technologists with the knowledge for this responsibility.
Risks & Costs of Inadequate Manufacturing Hygiene
Anyone involved in the manufacture of personal care products for any period of time has experienced the problem of a product failing its microbiological specification. There is no such thing as an inexpensive contamination event. More importantly, if the issue(s) are not addressed, the firm may be faced with a much more publicly visible manifestation of failed personal care manufacturing hygiene through FDA enforcement or EU RAPEX reports often expanded by social media. These are announced recalls of noncompliant, adulterated personal care products. Microbiological contamination is by far the most reported noncompliance.3 Such contamination is significant as contaminated personal care products in consumer use have been the source of infections. Whereas among healthy individuals, such infection may by superficial and transient, for those who are immunocompromised, living with comorbidities such as diabetes, cancer, or populations at the extremes of age, consequences can be severe. Many may consider this “at risk” population to be small, but estimates place this compromised group at 25% or more of the US population.4 Whereas the immunocompromised are at special risk, contaminated personal care products, especially eye area products, can impact healthy users in context of minor physical compromise.5 In other words, this is a very real concern for all personal care products manufacturers.Case 1
Fatal Infection from Contaminated ShampooOne tragic consequence of poor manufacturing hygiene was reported in Saudi Arabia.6 A baby shampoo heavily contaminated from manufacturing with Serratia marcescens was used to wash infants’ hair in a Rihad hospital. In context of naïve immunity, this common bacterium proved dangerous. Fifteen infants became seriously ill with conditions including bacteremia, septicemia, and respiratory and urinary tract infections, and one died. A similar report of fatal infection traced to a contaminated shampoo was reported among patients undergoing cancer chemotherapy treatment at the MD Anderson Hospital.7
Compromised microbiological quality can also prove dangerous to corporate survival as described by the following two case studies.
Case 2
Fungal Contamination from an Eye Care Product8,9A century-old, publicly-traded company with headquarters in New York state developed, and in 2005 introduced, a novel contact lens care product to the global market. Later that year, a report associating the product with potential fungal contamination and infection appeared in the literature. Increasing reports, especially in one Asian region, and investigations continued leading to a mid-2006 CDC publication associating the product with the reported infection stating, “nearly all of the company’s…products were linked to severe fungal eye infections.”
The product was subsequently recalled and removed from global markets. The investigation was extensive and its findings revealed significant harm was suffered by many users. The secondary effect on the company is also noteworthy. As expected, competitors assumed the company’s shelf space and market share. In response to the CDC report and FDA criticism that the company failed to report the previous international concerns, the stock market lost faith with a resulting 40% reduction in its market capitalization. Vulnerable to a buyout, the company was purchased in 2007 by a private equity (PE) firm with the announcement of a 25% workforce reduction. The PE firm relocated the company’s headquarters from its hometown of over a century.
In 2013, an international pharmaceutical company purchased the company from the PE firm, announced a 10-15% reduction in workforce and transferred its headquarters out of the US. The company settled lawsuits related to the issue with a payment over $200 million in 2013, and one can still find relevant attorney solicitations on the web. Although the origin of the fungal contamination remains obscure, a number of GMP violations were reported by government investigators concerning its manufacturing practices. However, the negative impacts on customers, company employees and fortune were clearly profound.
Case 3
Bacterial Contamination of a Hair Care ProductAnother well-established company, this one manufacturing hair care products, introduced a product combining highly effective technology and impactful marketing/advertising. These were so effective that market demand exceeded company expectations, to the extent that manufacturing capacity proved inadequate. Further burdened with multiple versions driven by the successful introduction, manufacturing looked for ways to increase production and found such questionable measures as decreased/eliminated cleaning sanitization between batches and versions, and reduced time of heat sanitization.
While the exact origin of the contamination is arguable, two reasonable culprits can be traced back to an unpreserved premix that slipped past the review process, and low-level contamination of the water system that was not adequately monitored. Regardless of the initial source, it was apparent that a biofilm had been established in the system that no level of cleaning or sanitization remediation could eliminate. With the detection of contamination, production was halted, and shipments were limited to compliant, warehoused product. Persistence of biofilm and contamination prevented further production causing supply issues.
With interruption of shipments, competitors’ products rapidly occupied the open retail shelves. Direct economic costs and less tangible opportunity costs of lost market momentum were substantial, and production was moved to a facility whose manufacturing hygiene was in control.
Some years later, the company’s acquisition of a competitor brought historic records indicating that one acquired product had been failing in the market at the time of the above-described issue. Unable to compete with the product discussed above, the former competitor was surprised at the opportunity to fill empty shelves that effectively rescued their failing product. Instead of the company’s technology and marketing acumen earning market dominance, failure in manufacturing hygiene allowed an inferior, competitive product to survive, evolve and grow, creating a market presence that the company later paid a significant price to acquire.
Where Are the Manufacturing Hygienists?
Manufacturing hygiene capabilities in some larger companies are served by highly-experienced and motivated technologists. These are classically-trained individuals who have gained ad hoc training due to their willingness to take this personal initiative. Through decades of practical experience, they established their expertise and credibility to effectively conduct manufacturing hygiene responsibilities. Such individuals are rare and not readily replaced. For many other companies, the function is addressed in isolated detail by multiple technologists managing an incomplete subset of quality elements.
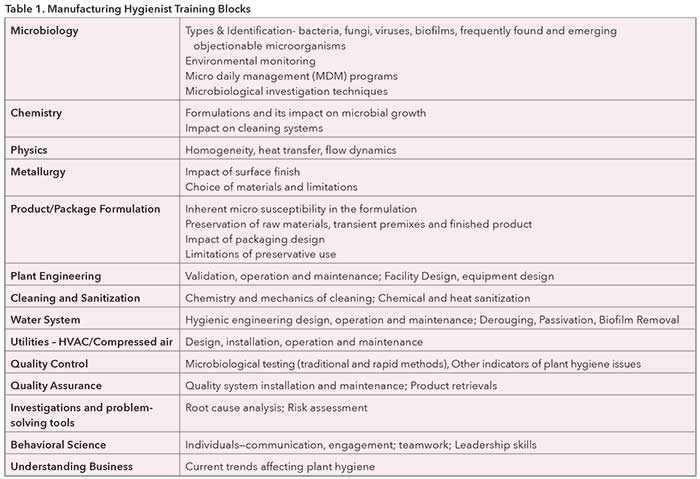
Industry experts from the task force, in collaboration with UC’s experts, will provide a complete curriculum addressing many aspects of cosmetic science and technology from manufacturing design through production. The planned curriculum will offer a Graduate Certificate in Manufacturing Hygiene with successful completion of 12-15 credit hours of relevant online courses. All courses will be available in an asynchronous format. The certificate program will be completed as a part-time study, and is therefore ideal for those who are already working in the field. In addition, provision also exists for continuation of the studies toward a Master’s degree in Cosmetic Science with focus on Manufacturing Hygiene. It will require a successful completion of additional 15-18 credit hours in UC’s current cosmetic curriculum. Discussed conceptually in Part II, the specific manufacturing hygiene training blocks being developed jointly by the UC and PCPC partnership are extensive (see Table 1). This remarkable collaboration will combine UC’s expertise in the development of cosmetic formulations with industry expertise in the manufacturing quality. Stay tuned for more information on this unique training opportunity.
Conclusion
In this three-part series, we defined the concept of the manufacturing hygienist as the conductor of quality. This capability is and will be increasingly pivotal to personal care product quality. We posit that as we continue to lose traditional preservative systems, historical issues of inadequate manufacturing hygiene will continue to be unmasked and exposed as an area of needed improvement. Having well-trained manufacturing hygienists will be critical to quickly addressing these issues. In order to ensure a steady pipeline of highly competent individuals to fill these manufacturing hygiene posts, PCPC is now partnering with the University of Cincinnati to provide appropriately trained and credentialed Manufacturing Hygienists equipped with the tools to conduct the industry’s quality – minimizing economic and supply chain disruptions, and most importantly, protecting the consumer’s health and safety.References:
- https://www.happi.com/issues/2022-03-01/view_experts-opinion/a-plant-hygienist-ensures-manufacturing-quality-product-integrity/?widget=listSection
- https://www.happi.com/issues/2022-04-01/view_experts-opinion/a-plant-hygienist-ensures-manufacturing-quality-am/?widget=listSection
- Wong, S., et al. 2000. Recalls of foods and cosmetics due to microbial contamination reported to the US Food and Drug Administration. Journal of Food protection, 63:1113-1116.
- Gerba, C.P., Rose, J.B. and Haas, C.N., 1996. Sensitive populations: who is at the greatest risk? International Journal of Food Microbiology, 30113-123.
- Wilson, L.A. and Ahearn, D.G., 1977. Pseudomonas-induced corneal ulcers associated with contaminated eye mascaras. American Journal of Ophthalmology, 84:112-119.
- Madani, T.A., et al., 2011. Serratia marcescens-contaminated baby shampoo causing an outbreak among newborns at King Abdulaziz University Hospital, Jeddah, Saudi Arabia. Journal of Hospital Infection, 78:16-19.
- Fainstein, V., et al. , 1988. Hair clipping: another hazard for granulocytopenic patients? Journal of Infectious Diseases, 158:655-656.
- Janetos, T.M. and Thyparampil, P., 2019. Ocular Cosmetics: Public Safety Problem or Harmless Products? Ophthalmic Plastic & Reconstructive Surgery, 35:294-295.
- Epstein, A.B., 2007. In the aftermath of Fusarium fungal keratitis outbreak: What have we learned? Clinical Ophthalmology 1:255-266.
- https://online.uc.edu/masters-programs/ms-in-cosmetic-science/













