Nadim Shaath, Alpha Research & Development Ltd11.30.22
Nearly half a century ago, the Food & Drug Administration (FDA) issued its first regulations (Advanced Notice of Proposed Rulemaking in 1978) and approved 21 UV filters that were designated as Category I. Only a handful of these UV filters survived the test of time, thankfully supported by three recently approved filters by the FDA, mostly designed by European scientists, namely avobenzone (Givaudan), zinc oxide (BASF and others) and Ecamsule (L’Oréal).
European researchers march on and brought to market superior UV filters that are mostly based on the 500 Dalton rule. This increased molecular weight in UV filters has added the necessary conjugated molecules that produce better protection in the longer wavelength UVA radiation. These molecules are sorely needed for skin cancer protection. They allow for broad spectrum protection, higher extinction coefficients (use less in sunscreen product), and reduce the chance of permeability in the skin, thereby increasing the safety of these molecules. The Europeans have also introduced more than a half dozen other superior UVA filters that, for years, we have been calling for their approval in the US through the FDA Time and Extent Application (TEA) process, but to no avail! These European blockbuster UVA filters that have been used freely worldwide for many years include Tinosorb S, Tinosorb M, Mexoryl XL, Uvinul A Plus and others.
If Hawaii has its way and oxybenzone, octinoxate and other so-called chemical filters are eliminated as approved filters, we are left with only zinc oxide, ecamsule, avobenzone that have some protection in the UVAII region (320-350 nm) and a few others in the US. Zinc oxide has its problems with whitening and SPF rating. Ecamsule is patent protected by L’Oréal and is water soluble. Avobenzone is notorious for its photo-instability requiring quenchers and photo-stabilizers. Interestingly, the first photo-stabilization of avobenzone was introduced by a L’Oréal patent in its use of octocrylene to solve this photo-instability issue.
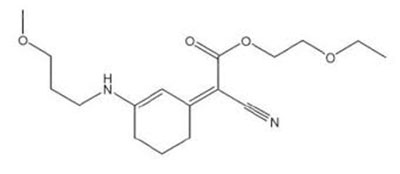
L’Oréal’s Mexoryl 400
Meanwhile, several new filters are being released in Europe by L’Oréal, BASF and Pierre Fabre. This year, L’Oréal introduced a UVAI (340-400nm) filter that received approval last year by the Scientific Committee on Consumer Safety (SCCS) in Europe. This molecule, commercially called Mexoryl 400, was assigned the COLIPA code S87 (safe at 5%) and can be used worldwide except in the US and a few other countries around the world that follow US FDA regulations. In fact, a full line of sunscreen products under the brand name La Roche-Posay were introduced in the market in May. This molecule has a 60,000 Extinction Coefficient (compared to homosalate at 4,500!!) and has a broad-spectrum range protection from 300 to 400nm! The INCI name is Methoxypropylamino cyclohexenylidene ethoxyethylcyanoacetate (MCE). Its CAS number is 1419401-88-9, and its EC number is 700-860-3. Despite its rather long chemical name and an empirical formula of C17H26N2O4, MCE interestingly has a molecular weight of only 322. Its chemical structure is shown at left.
Do we not have superior scientists in the US? Of course we do. The problem is simply that FDA introduced multiple barriers and raised the bar for approval of new filters, all leading to increased costs, additional time and prohibitive drug status requirements. FDA did not provide the economic incentive to allow US companies to spend the time and money needed to invest in superior UV protection. In the past half century, the FDA has not allowed a single new UV filter to be approved in the US. The only exceptions are the three European ingredients listed earlier in this article.
At the end of the day, of course, it does not matter whether it’s American scientists or European scientists or Japanese scientists that provide us with superior UV filters and better protection, but it still hurts that we were not freed of the FDA restrictions in exploring new ingredients. American scientists’ contributions in introducing superior drugs and protocols to prevent and contain skin cancer have been remarkable.
Reductions and reversals of melanomas and skin cancers have improved significantly. American scientists still lead the way in the creation and production of unique cosmetic formulations used in sunscreens. Americans improved the water resistance, the feel and the ease of application of sunscreen products, too. Our scientists were the leaders in setting the conditions and parameters of SPF testing and protocols; unfortunately, we failed in creating effective UV molecules—the heart and soul of the sunscreen product—that better protect us from the ravishing UV and total solar radiation.
I am not advocating the use of Mexoryl 400 as a panacea, but it’s a step in the right direction. If we want to safely enjoy the sun, we need to address the issue of better UV filters. A few innovations have surfaced in the US, including superior quenchers, photo-stabilizers, and SPF boosters. Notably, Delavie Sciences introduced an ingredient that was created by NASA scientists. It is totally natural, safe, offers SPF boosting capabilities and has antioxidant properties, thereby assisting in better protection from UV, HEV and IR radiation. This ingredient could linger “in space” without a real home if the FDA does not ease its restrictions in regulations and allow unique innovations from American ingenuity to come to fruition.

Environmental protection is important, too.
Congress should get involved in finding ways to offer incentives to protect, promote and enhance American ingenuity in creating superior solar protection. FDA should work closely with American scientists, entrepreneurs, American universities and colleges, and cosmetic and UV ingredient companies to encourage research in this field and to monitor the process leading to the adoption of superior UV and solar filters for protection. FDA should reassert its regulatory role in arbitrating and being the only body that sets practical rules and regulations in sunscreens and not allow states or individuals to independently approve or ban UV filters for local issues and concerns.
Of course, environmental protection is paramount, and for that I hail the recent National Academy of Sciences (NAS) study that was just concluded highlighting the need for superior sunscreen protection to reduce skin cancers and that the so-called chemical UV filters are not yet proven to be the real villains in coral reef damage. This current wave of producing the so-called Reef Safe filters has led to several UV filter bans with no consequences given to the reduced solar protection from the hazardous rays to the consumer. NAS’s work is obviously incomplete as it called on the Environmental Protection Agency to study the ecological risk assessment (ERA) for all currently marketed UV filters. We should accept the principle of risk and benefit ratio now applied in medicine. The pharmaceutical industry is allowed to introduce and market drugs for cancer, for reducing blood pressure, for lowering cholesterol and ultimately preventing heart attacks; yet every drug includes warnings of serious side effects—including death—if you take those drugs. The side effects of sunscreen use to the environment and individuals are minimal, especially if they are better designed than some of the currently approved US filters.
Perhaps it is time to reconsider changing the status of sunscreens as OTC drugs to cosmetics and to also not allow the use of extreme terms such as “the use of sunscreens reduces skin cancers.” Sunscreens of the future should be designed so that they are broad spectrum, more efficient, safer, more natural and non-permeable into the skin. Their role is not to reverse or protect from skin cancer; rather, their role is only to shield the individual from receiving the harmful solar radiation directly on their skin, period. Their protection should be similar to an umbrella, clothing or even a shade tree.
As I look back at US sunscreen developments, I am reminded of the Joan Baez song, “Time Is Passing Us By.” It was released around the time the initial FDA regulations were introduced in 1978, Baez’s last line provides us with a glimmer of hope: “And I’m good for one more try.”
At this point in time, I think it’s time for one more try.
Nadim Shaath
Alpha Research & Development Ltd
Nadim@alpharnd.com
Dr. Nadim Shaath is the president of Alpha Research & Development, Ltd. in White Plains, NY. He has over 30 years of experience in the sunscreen industry. He served as the Chairman of the chemistry department at SUNY-Purchase and the CEO of Kato Worldwide. Recently he published his new book entitled “Healing Civilizations: The Search for Therapeutic Essential Oils and Nutrients” Cameron Books, Petaluma, CA.
European researchers march on and brought to market superior UV filters that are mostly based on the 500 Dalton rule. This increased molecular weight in UV filters has added the necessary conjugated molecules that produce better protection in the longer wavelength UVA radiation. These molecules are sorely needed for skin cancer protection. They allow for broad spectrum protection, higher extinction coefficients (use less in sunscreen product), and reduce the chance of permeability in the skin, thereby increasing the safety of these molecules. The Europeans have also introduced more than a half dozen other superior UVA filters that, for years, we have been calling for their approval in the US through the FDA Time and Extent Application (TEA) process, but to no avail! These European blockbuster UVA filters that have been used freely worldwide for many years include Tinosorb S, Tinosorb M, Mexoryl XL, Uvinul A Plus and others.
If Hawaii has its way and oxybenzone, octinoxate and other so-called chemical filters are eliminated as approved filters, we are left with only zinc oxide, ecamsule, avobenzone that have some protection in the UVAII region (320-350 nm) and a few others in the US. Zinc oxide has its problems with whitening and SPF rating. Ecamsule is patent protected by L’Oréal and is water soluble. Avobenzone is notorious for its photo-instability requiring quenchers and photo-stabilizers. Interestingly, the first photo-stabilization of avobenzone was introduced by a L’Oréal patent in its use of octocrylene to solve this photo-instability issue.
New Advances

L’Oréal’s Mexoryl 400
Do we not have superior scientists in the US? Of course we do. The problem is simply that FDA introduced multiple barriers and raised the bar for approval of new filters, all leading to increased costs, additional time and prohibitive drug status requirements. FDA did not provide the economic incentive to allow US companies to spend the time and money needed to invest in superior UV protection. In the past half century, the FDA has not allowed a single new UV filter to be approved in the US. The only exceptions are the three European ingredients listed earlier in this article.
At the end of the day, of course, it does not matter whether it’s American scientists or European scientists or Japanese scientists that provide us with superior UV filters and better protection, but it still hurts that we were not freed of the FDA restrictions in exploring new ingredients. American scientists’ contributions in introducing superior drugs and protocols to prevent and contain skin cancer have been remarkable.
Reductions and reversals of melanomas and skin cancers have improved significantly. American scientists still lead the way in the creation and production of unique cosmetic formulations used in sunscreens. Americans improved the water resistance, the feel and the ease of application of sunscreen products, too. Our scientists were the leaders in setting the conditions and parameters of SPF testing and protocols; unfortunately, we failed in creating effective UV molecules—the heart and soul of the sunscreen product—that better protect us from the ravishing UV and total solar radiation.
I am not advocating the use of Mexoryl 400 as a panacea, but it’s a step in the right direction. If we want to safely enjoy the sun, we need to address the issue of better UV filters. A few innovations have surfaced in the US, including superior quenchers, photo-stabilizers, and SPF boosters. Notably, Delavie Sciences introduced an ingredient that was created by NASA scientists. It is totally natural, safe, offers SPF boosting capabilities and has antioxidant properties, thereby assisting in better protection from UV, HEV and IR radiation. This ingredient could linger “in space” without a real home if the FDA does not ease its restrictions in regulations and allow unique innovations from American ingenuity to come to fruition.
What Must Be Done?

Environmental protection is important, too.
Of course, environmental protection is paramount, and for that I hail the recent National Academy of Sciences (NAS) study that was just concluded highlighting the need for superior sunscreen protection to reduce skin cancers and that the so-called chemical UV filters are not yet proven to be the real villains in coral reef damage. This current wave of producing the so-called Reef Safe filters has led to several UV filter bans with no consequences given to the reduced solar protection from the hazardous rays to the consumer. NAS’s work is obviously incomplete as it called on the Environmental Protection Agency to study the ecological risk assessment (ERA) for all currently marketed UV filters. We should accept the principle of risk and benefit ratio now applied in medicine. The pharmaceutical industry is allowed to introduce and market drugs for cancer, for reducing blood pressure, for lowering cholesterol and ultimately preventing heart attacks; yet every drug includes warnings of serious side effects—including death—if you take those drugs. The side effects of sunscreen use to the environment and individuals are minimal, especially if they are better designed than some of the currently approved US filters.
Perhaps it is time to reconsider changing the status of sunscreens as OTC drugs to cosmetics and to also not allow the use of extreme terms such as “the use of sunscreens reduces skin cancers.” Sunscreens of the future should be designed so that they are broad spectrum, more efficient, safer, more natural and non-permeable into the skin. Their role is not to reverse or protect from skin cancer; rather, their role is only to shield the individual from receiving the harmful solar radiation directly on their skin, period. Their protection should be similar to an umbrella, clothing or even a shade tree.
As I look back at US sunscreen developments, I am reminded of the Joan Baez song, “Time Is Passing Us By.” It was released around the time the initial FDA regulations were introduced in 1978, Baez’s last line provides us with a glimmer of hope: “And I’m good for one more try.”
At this point in time, I think it’s time for one more try.
Nadim Shaath
Alpha Research & Development Ltd
Nadim@alpharnd.com
Dr. Nadim Shaath is the president of Alpha Research & Development, Ltd. in White Plains, NY. He has over 30 years of experience in the sunscreen industry. He served as the Chairman of the chemistry department at SUNY-Purchase and the CEO of Kato Worldwide. Recently he published his new book entitled “Healing Civilizations: The Search for Therapeutic Essential Oils and Nutrients” Cameron Books, Petaluma, CA.













