08.30.18
Fiscal 2018 was a great for Estee Lauder Companies (ELC), as the firm earlier this month reported net sales rose 16% to $13.68 billion.
According to Fabrizio Freda, president and CEO, ELC generated higher sales in every region and product category and gained global share.
“By investing in our hero franchises, fast-growing channels and digital and social media, we delivered double-digit sales and adjusted earnings per share growth. We achieved record net sales in fiscal 2018 and one of our best performances in the last decade," he said when the company's fiscal year results were released this summer.
What’s helped get ELC to this point? While social media and some great colors no doubt are part of it, investment in science plays a role, too. According to the company’s own data, ELC’s research and development costs were $179 million in 2017. While that was a drop from the $191 million spent in fiscal 2016, it was on par with levels reported for fiscal 2015.
So just what has R&D been working on out in Melville? Here’s a look at five of ELC’s most recently awarded patents:
MudMask with Real Leaf
US Patent No. 9,987,218 B2; ELC Management was awarded patent protection for this method of making a packaged leaf mud mask product. The steps to form the leaf mud are: separating a leaf batch of whole leaves into three sub-batches, a first sub-batch, a second sub-batch, and a third sub-batch; grinding the first sub-batch into a fine powder; chopping the third sub-batch into partially chopped leaves; heating water; adding the fine powder and second sub-batch to the heated water for a period of time to form a brewed tea; adding, after the period of time, the partially chopped leaves and mud to the brewed tea to form a mud solution; and
mixing the mud solution to form the leaf mud mask product.
Treating Skin for Inflammation; Screening for Actives to Stimulate Pro-Resolution Pathways
US Patent No. 10,010,492 B2; ELC Management LLC earned a patent for a method for formulating a topical composition comprising:
(A) Identifying an inflammatory metabolite Inhibitor by exposing cells treated and untreated with a test ingredient to an inflammation precipitating condition, measuring the amount of inflammatory metabolites or inflammatory metabolite markers released and selecting the test ingredient that causes a decrease in cellular release of Inflammatory metabolites or inflammatory metabolite markers when compared to untreated cells wherein: the inflammatory metabolites are one or more of cyclic endoperoxidases derived from arachidonic acid; prostacyclin 12 (PG12): prostaglandin E2 (PGE2); prostaglandin F2 alpha (PGF2 alpha); prostaglandin A2 (PGA2); prostaglandin D2 (PGD2); leukotriene A4 (LTA4): leukotriene B4 (LTB4); leukotriene C4 (LTC4); leukotriene D4 (LTD4); platelet activating factor (PAF); interleukin-1 alpha (IL-1 alpha); interleukin-1 beta (IL-1 beta); interleukin-6 (IL-6); interleukin-8 (IL-8); tumor necrosis factor alpha (TNF alpha); and monocyte chemotactice protein (MCP-1); and the inflammatory metabolite markers are one or more of 5-hydroxyeicosatetraenoic acid (5-HETE); 5-hydroxyperoxyeicosatetraenoic acid (H-HPETE); leukotriene E4 (LTE4); prostaglandin G2 (PGG2); prostaglandin H2 (PGH2); endoperoxide precursors of PGE2; prostaglandin J2 (PGJ2); prostaglandin 12 (PG12); and 6-keto prostaglandin F1 alpha;
(B) Identifying a pro-resolving activator by exposing cells treated and untreated with a test ingredient to an inflammation precipitating condition, measuring the amount of pro-resolving activators released and selecting the test ingredient that shows an increase in cellular release of pro-resolving activators when compared to the untreated cells wherein the pro-resolving activator is one or more of a pro-resolving lipid mediator which is one or more of resolvin; protectin; lipoxin or maresin; or a pro-resolving lipid mediator marker which is one or more of cyclooxygenase (COX); lipoxygenase (LOX); cytochrome epoxygenase (CYPe); cyctochrome hydrolase (CYP); 15-hydroxyeicosapentaenoic acid (15-HETE); 12-hydroxyeicosapentaenoic acid (12-HETE); 14- hydroxydocosahexaenoic acid (14-HDOHE); 18-hydroxyeicosapentaenoic acid (18-HEPE); and 17-hydroxydocosahexaenoic acid (17HDOHE); and;
(C) Formulating the test ingredient(s) selected in (a) and (b) into a topical composition.
Stabilizing Riboflavin
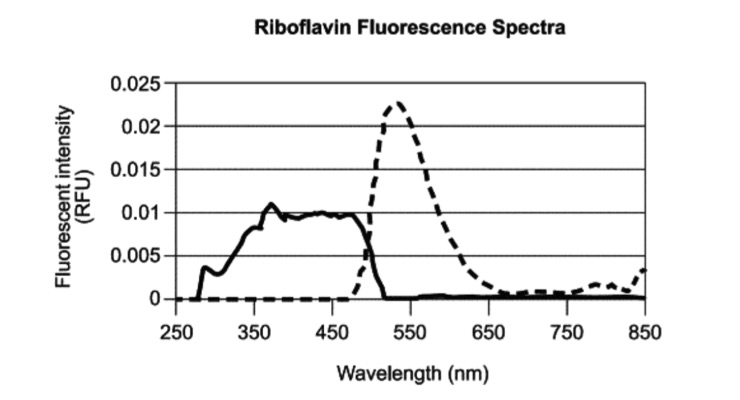
US Patent No. 10,030,022 B2; ELC Management LLC, Melville, NY, has patented a method for stabilizing riboflavin against degradation upon exposure to UV light by mixing a silicone polyamide substrate with a solution of riboflavin, and evaporating the liquid to form an optically-activated complex in the form of a gel that will absorb light in the wavelength range of 260 to 460nm and emit light in the wavelength range of 470 to 650nm.
Composition with SIRT6 Activating Peptide
US Patent No. 10,022,314 B2; ELC Management has patented a composition that comprises 0.0001% to 1% of a SIRT6 activating peptide and 0.0001%-0.05% of 8-Oxoguanine glycosylase, by weight of the composition. The SIRT6 activating peptide is incorporated into the composition in the form of yeast ferment extract.
Method, Composition To Improve Skin Appearance
US Patent No. 9,750,682 B2; ELC Management patented a method for stimulating collagen synthesis in skin cells in need of such treatment by topically applying a composition comprising at least one extract from Laminaria genus, at least one extract from the Narcissus genus, and at least one peptide of the formula (I):
R1-(AA)n-X1-X2-X3-X4-X5-X6-(AA)p-R2, in which
X1 is glycine or threonine or histidine;
X2 is alanine or glutamine or glycine;
X3 is glycine or asparagine or serine;
X4 is valine or isoleucine or leucine;
X5 is serine or aspartic acid or phenylalanine;
X6 is alanine or glutamic acid or lysine; wherein
when X1 is glycine, then X2 is alanine and X3 is glycine;
when X1 is threonine, then X3 is asparagine;
when X1 is histidine, then X2 is glycine;
AA represents any amino acid and n and p are integers between 0 and 2;
R1 represents the primary amino function of the N-terminal amino acid, free or substituted by an acyl type group having either an alkyl chain from C1-30, saturated or unsaturated, that may be an acetyl group, or an aromatic group that may be chosen from among a benzoyl, tosyl or benzyloxycarbonyl type group; and
R2 represents the hydroxyl group of the carboxyl function of the C-terminal amino acid, free or substituted by a group that may be chosen from among an alkyl chain from C1-30, or an NH2, NHY or NYY group with Y representing an alkyl chain from C1-4 wherein the extract from the Laminaria genus is present at 0.001 to 1%, the extract from Narcissus genus is present at 0.001 to 1.5% and the peptide is present from 0.0001 to 1%, with all percentages by weight of the total composition.
Looking for more recently filed patents? Our Patent News page can be found here.
According to Fabrizio Freda, president and CEO, ELC generated higher sales in every region and product category and gained global share.
“By investing in our hero franchises, fast-growing channels and digital and social media, we delivered double-digit sales and adjusted earnings per share growth. We achieved record net sales in fiscal 2018 and one of our best performances in the last decade," he said when the company's fiscal year results were released this summer.
What’s helped get ELC to this point? While social media and some great colors no doubt are part of it, investment in science plays a role, too. According to the company’s own data, ELC’s research and development costs were $179 million in 2017. While that was a drop from the $191 million spent in fiscal 2016, it was on par with levels reported for fiscal 2015.
So just what has R&D been working on out in Melville? Here’s a look at five of ELC’s most recently awarded patents:
MudMask with Real Leaf
US Patent No. 9,987,218 B2; ELC Management was awarded patent protection for this method of making a packaged leaf mud mask product. The steps to form the leaf mud are: separating a leaf batch of whole leaves into three sub-batches, a first sub-batch, a second sub-batch, and a third sub-batch; grinding the first sub-batch into a fine powder; chopping the third sub-batch into partially chopped leaves; heating water; adding the fine powder and second sub-batch to the heated water for a period of time to form a brewed tea; adding, after the period of time, the partially chopped leaves and mud to the brewed tea to form a mud solution; and
mixing the mud solution to form the leaf mud mask product.
Treating Skin for Inflammation; Screening for Actives to Stimulate Pro-Resolution Pathways
US Patent No. 10,010,492 B2; ELC Management LLC earned a patent for a method for formulating a topical composition comprising:
(A) Identifying an inflammatory metabolite Inhibitor by exposing cells treated and untreated with a test ingredient to an inflammation precipitating condition, measuring the amount of inflammatory metabolites or inflammatory metabolite markers released and selecting the test ingredient that causes a decrease in cellular release of Inflammatory metabolites or inflammatory metabolite markers when compared to untreated cells wherein: the inflammatory metabolites are one or more of cyclic endoperoxidases derived from arachidonic acid; prostacyclin 12 (PG12): prostaglandin E2 (PGE2); prostaglandin F2 alpha (PGF2 alpha); prostaglandin A2 (PGA2); prostaglandin D2 (PGD2); leukotriene A4 (LTA4): leukotriene B4 (LTB4); leukotriene C4 (LTC4); leukotriene D4 (LTD4); platelet activating factor (PAF); interleukin-1 alpha (IL-1 alpha); interleukin-1 beta (IL-1 beta); interleukin-6 (IL-6); interleukin-8 (IL-8); tumor necrosis factor alpha (TNF alpha); and monocyte chemotactice protein (MCP-1); and the inflammatory metabolite markers are one or more of 5-hydroxyeicosatetraenoic acid (5-HETE); 5-hydroxyperoxyeicosatetraenoic acid (H-HPETE); leukotriene E4 (LTE4); prostaglandin G2 (PGG2); prostaglandin H2 (PGH2); endoperoxide precursors of PGE2; prostaglandin J2 (PGJ2); prostaglandin 12 (PG12); and 6-keto prostaglandin F1 alpha;
(B) Identifying a pro-resolving activator by exposing cells treated and untreated with a test ingredient to an inflammation precipitating condition, measuring the amount of pro-resolving activators released and selecting the test ingredient that shows an increase in cellular release of pro-resolving activators when compared to the untreated cells wherein the pro-resolving activator is one or more of a pro-resolving lipid mediator which is one or more of resolvin; protectin; lipoxin or maresin; or a pro-resolving lipid mediator marker which is one or more of cyclooxygenase (COX); lipoxygenase (LOX); cytochrome epoxygenase (CYPe); cyctochrome hydrolase (CYP); 15-hydroxyeicosapentaenoic acid (15-HETE); 12-hydroxyeicosapentaenoic acid (12-HETE); 14- hydroxydocosahexaenoic acid (14-HDOHE); 18-hydroxyeicosapentaenoic acid (18-HEPE); and 17-hydroxydocosahexaenoic acid (17HDOHE); and;
(C) Formulating the test ingredient(s) selected in (a) and (b) into a topical composition.
Stabilizing Riboflavin
US Patent No. 10,030,022 B2; ELC Management LLC, Melville, NY, has patented a method for stabilizing riboflavin against degradation upon exposure to UV light by mixing a silicone polyamide substrate with a solution of riboflavin, and evaporating the liquid to form an optically-activated complex in the form of a gel that will absorb light in the wavelength range of 260 to 460nm and emit light in the wavelength range of 470 to 650nm.

Composition with SIRT6 Activating Peptide
US Patent No. 10,022,314 B2; ELC Management has patented a composition that comprises 0.0001% to 1% of a SIRT6 activating peptide and 0.0001%-0.05% of 8-Oxoguanine glycosylase, by weight of the composition. The SIRT6 activating peptide is incorporated into the composition in the form of yeast ferment extract.
Method, Composition To Improve Skin Appearance
US Patent No. 9,750,682 B2; ELC Management patented a method for stimulating collagen synthesis in skin cells in need of such treatment by topically applying a composition comprising at least one extract from Laminaria genus, at least one extract from the Narcissus genus, and at least one peptide of the formula (I):
R1-(AA)n-X1-X2-X3-X4-X5-X6-(AA)p-R2, in which
X1 is glycine or threonine or histidine;
X2 is alanine or glutamine or glycine;
X3 is glycine or asparagine or serine;
X4 is valine or isoleucine or leucine;
X5 is serine or aspartic acid or phenylalanine;
X6 is alanine or glutamic acid or lysine; wherein
when X1 is glycine, then X2 is alanine and X3 is glycine;
when X1 is threonine, then X3 is asparagine;
when X1 is histidine, then X2 is glycine;
AA represents any amino acid and n and p are integers between 0 and 2;
R1 represents the primary amino function of the N-terminal amino acid, free or substituted by an acyl type group having either an alkyl chain from C1-30, saturated or unsaturated, that may be an acetyl group, or an aromatic group that may be chosen from among a benzoyl, tosyl or benzyloxycarbonyl type group; and
R2 represents the hydroxyl group of the carboxyl function of the C-terminal amino acid, free or substituted by a group that may be chosen from among an alkyl chain from C1-30, or an NH2, NHY or NYY group with Y representing an alkyl chain from C1-4 wherein the extract from the Laminaria genus is present at 0.001 to 1%, the extract from Narcissus genus is present at 0.001 to 1.5% and the peptide is present from 0.0001 to 1%, with all percentages by weight of the total composition.
Looking for more recently filed patents? Our Patent News page can be found here.