Amirah Bajawi, Cameron Whitney and Dr. Teresa Berninger, Jungbunzlauer 12.02.20
Formulators must consider a number of factors when developing cosmetic products. This is no different for products related to hair care. From shampoos and conditioners, to styling products—a delicate balance exists between formulating for performance and market trend awareness. The growing demand for specific free-from statements as well as the need for more bio-based ingredients creates challenges in product development and marketing.
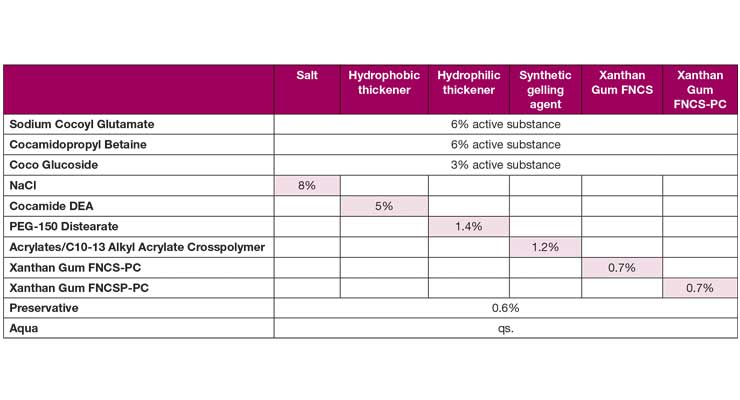
Table 1: Comparison of different thickeners in natural, sulfate-free formulation
When it comes to shampoos, the most important property is rheology, which has a dominant influence on how the consumer perceives the product. Many thickeners can influence the rheology, from salts to complex polymers, but when it comes to consumer safety and perception, traditional thickeners like PEG derivatives or cocamide DEA have fallen out of favor. When thinking about other solutions, one should also consider the “microplastic” factor. Although widely-banned exfoliating microbeads may first come to mind, “microplastics” regulations could one day impact commonly-used soluble polymers, specifically those that do not readily degrade in the environment.1 Consumers may also have a negative perception of these materials. Fortunately, there are a number of readily biodegradable polymer solutions, including naturally-occurring polymers like starches or gums. Xanthan gum is of particular interest because it possesses many qualities uniquely suited for thickening shampoos.
Xanthan gum is a polymer of repeating sugar units somewhat similar to cellulose. But unlike cellulose, xanthan gum possesses a trisaccharide moiety present along the backbone of the molecule, repeating every other glycosyl residue. Thanks to its distinctive structure, xanthan gum provides a host of benefits when used as a thickener. It provides excellent thickening even at low concentrations. It is resilient to pH variations; therefore, it is stable in both alkaline and acidic conditions. It also possesses very good thermal stability so it can be easily added into hot or cold processes and it is compatible with a wide variety of other ingredients. In shampoos, it exhibits very good compatibility with surfactants and has shown positive effects on foam formation, height and stability.2
When choosing surfactants, one must consider the move toward sulfate-free varieties to create milder formulations. In the context of natural ingredients, there also exist a number of bio-based options with great potential. With any new class of ingredients come challenges in how they may impact performance or interact with other raw materials. For example, traditional thickening of formulations by adding salt is not feasible with some bio-based surfactants. The aim of this study was to see if the unique qualities of xanthan gum could benefit formulators looking to utilize these surfactants. One consideration with xanthan gum is that standard grades may not be able to replace traditional thickeners without compromising the appearance, texture or feel of the product. By tweaking the processing, unique grades of xanthan gum with reduced pseudoplasticity were developed to alleviate these potential sensory issues. We will explore such a grade by highlighting its utility in a sulfate-free shampoo.
Results & Discussions
In order to better understand the behavior of a reduced pseudoplastic xanthan gum with surfactants, a variety of nonionic, anionic and amphoteric surfactants were dissolved into a 1% w/w xanthan gum solution at varying concentrations. After dissolution, the pH was adjusted to 5.5, and the resulting samples were monitored for one month. During this time, optical evaluations and viscosity measurements were made. Glycolipids, coco-glucosides, caprylol/caproyl methyl glucamide, sodium cocoyl glutamate and cocamidopropyl betaine were among the range of sulfate-free, bio-based surfactants tested. Since it is rare that only single surfactant systems are used in formulations, dual-combinations of the surfactants were also dissolved into 1% xanthan gum solutions. An active concentration of 12% was chosen for each surfactant combination. Further, a 1:1 ratio was selected when it was not possible to categorize main- and co-surfactants. If categorization was possible, a ratio of 5:1 was tested. All of the surfactants and surfactant blends exhibited excellent stability in combination with xanthan gum.
The rheological behavior is important to characterize the shampoo application due to its wide influences on the flow of the shampoo out of the bottle, the behavior on the hand, how well it distributes into the hair and even storage stability.
To measure the flow curve at 21°C, a Haake Rheostress rheometer with cone-plate geometry (C35 / 2°Ti L) was used. The apparent viscosity was measured by an increasing shear rate from 0.01 to 100 s-1. The storage and the behavior on the hand after application are described by a low shear rate. A medium shear rate represents the application on the hair. Squeezing the shampoo out of the bottle is represented by a high shear rate.3,4
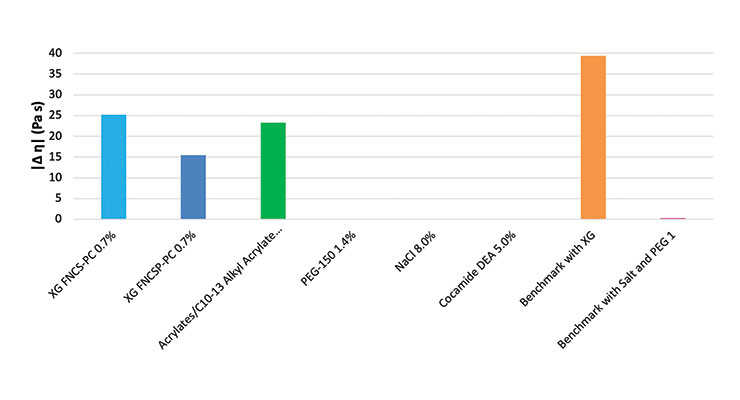
As a first step, different benchmarks were measured to define representative target values of viscosity (Fig. 1). Depending on the thickener, different flow behaviors were observed. The benchmark, thickened with a combination of salt and PEG, showed Newtonian behavior, which means with an increasing shear rate the apparent viscosity remained constant. The apparent viscosity of the benchmark thickened with xanthan gum decreased with an increasing shear rate, so the benchmark containing xanthan gum showed shear-thinning behavior.5
To understand the rheological behavior of different thickeners with natural surfactants, thickeners were combined with a typical, natural surfactant system. This system included a combination of sodium cocoyl glutamate, cocamidopropyl betaine and coco glucoside in a 2:2:1 ratio based on active substance. The total concentration of this system was 12% w/w, also based on active substance. The aim was to come as close to the apparent viscosity defined by the benchmarks
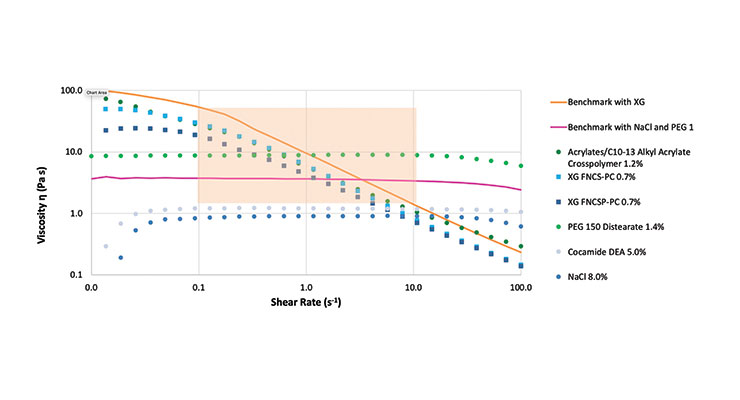
Figure 1: Rheology results
as possible (orange box in Fig. 1). The apparent viscosity should be around 10 Pa·s at a shear rate of 1 s-1.
Thickening via salt addition was the first approach evaluated since it is the traditional way of increasing viscosity. A hydrophobic thickener (cocamide DEA), a hydrophilic thickener (PEG-150 distearate) and a synthetic gelling agent (acrylates/C10-13 alkyl acrylate crosspolymer) were also tested to compare the rheological behavior to a surfactant system thickened with xanthan gum. As mentioned previously, there are multiple grades of xanthan gum each with different flow behaviors. For this study we looked at two clear solution grades, the first one with a standard flow behavior (FNCS-PC) and the second one with a reduced pseudoplasticity (FNCSP-PC). The amount of thickening agent was adapted to the rheological target values from the benchmark. The formulation is shown in Table 1.
The flow curves are shown in Fig. 1. The variability in flow behavior results from different thickening mechanisms. Salt and cocamide DEA are promoting the arrangement of surfactants into rod-shaped micelles, forming a thickening network with Newtonian behavior. Also the surfactant system thickened with PEG-150 distearate exhibits Newtonian flow behavior due to the crosslinking of the micelles and the increase of their size. In contrast, xanthan gum and the synthetic gelling agent (acrylates/C10-13 alkyl acrylate crosspolymer) are thickening the aqueous phase, which leads to shear thinning behavior of the thickened surfactant system.6
Our model system of natural and sulfate-free surfactants could not be thickened with salt or cocamide DEA. The viscosity did not reach the defined target even with elevated concentrations of salt which highlights the problem formulators face trying to thicken natural, sulfate-free surfactant systems. The traditional approach of thickening with salt does not work. The desired viscosity can be reached by using PEG-150 distearate; however, PEG derivatives may be undesirable from a consumer point of view, since they facilitate penetration of potentially harmful substances through the skin.7 Xanthan gum and the synthetic gelling agent (acrylates/C10-13 alkyl acrylate crosspolymer) show the desired viscosity. However, as awareness for synthetic polymers rises, the synthetic gelling agent (acrylates/C10-13 alkyl acrylate crosspolymer) may be viewed by the consumers as “microplastic.”1
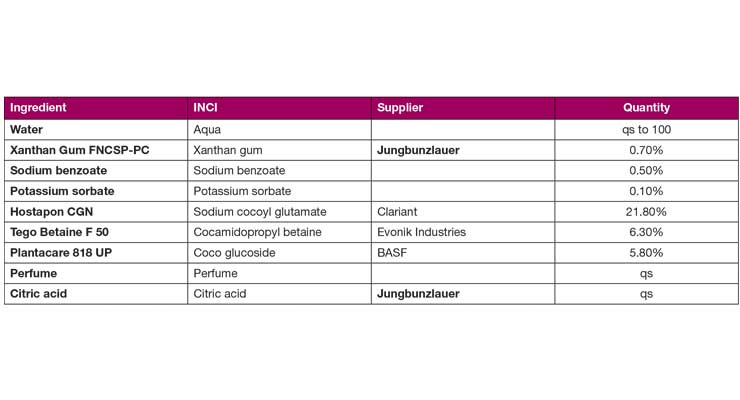
Table 2. Xanthan gum thickened sulfate-free shampoo (pH adjusted to 5.2-5.7).
To show the differences of the slopes, a comparison of the slope between 0.1 and 10 s-1 is shown on the graph above Fig. 1. As previously noted, Newtonian flow behavior means that the viscosity is independent from the shear rate, this means that the slope is about zero. Because of this, the surfactant systems thickened with salt, PEG-150 distearate and cocamide DEA have no slope. The advantage of xanthan gum with reduced pseudoplasticity (FNCSP-PC) is clearly visible in Figure 1. The slope is much smaller than that of the standard xanthan gum (FNCS-PC) and the synthetic gelling agent (acrylates/C10-13 alkyl acrylate crosspolymer). The shear thinning is less pronounced. In consequence, flow properties can be optimized to more closely resemble a benchmark with a traditional thickening system consisting of salt.
Discussion of Sensory Data
Sensory testing was performed to see just how close a reduced pseudoplastic xanthan gum could come to feeling like a traditionally thickened formulation. A group of 12 trained panelists was asked to compare benchmark, commercially available sulfate-free shampoo thickened with PEG and salt to a sulfate-free formulation successfully thickened with xanthan gum FNCSP-PC. Table 2 describes the test formulation in detail.
The panelists were given two shampoos and asked to compare the controllability of dosing, viscosity and haptics. In these three particular categories, a preference was indicated. Based on the results shown in Fig. 2, there was not a significant difference in the preference between the benchmark shampoo and the xanthan gum thickened sulfate-free formulation. Thus, we can conclude that xanthan gum FNCSP-PC
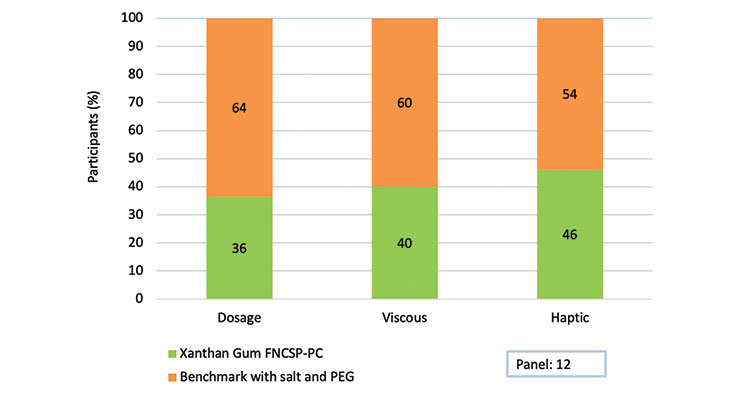
Figure 2. Results of the sensory evaluation.
helps enerate a similarly pleasant user experience in combination with bio-based, sulfate-free surfactants.
Conclusion
While most thickeners may work just fine in combination with surfactants like SLS and SLES in shampoo formulations, new regulations and the move toward more bio-based surfactants may pose significant challenges. Based off the stability studies, rheological measurements, and sensory evaluation, it is clear that one can effectively thicken in the presence of bio-based, sulfate-free surfactants without compromising the feel of the product using xanthan gum. In addition, xanthan gum is appropriate for natural formulations. This data demonstrates that xanthan gum is a powerful tool for formulators looking to clean up their labels.
More information
Amirah Bajawi, application technology assistant, Jungbunzlauer Ladenburg GmbH, Email: amirah.bajawi@jungbunzlauer.com; Cameron Whitney, market development manager, Jungbunzlauer Inc.; Email: cameron.whitney@jungbunzlauer.com; Dr. Teresa Berningers, senior project manager application technology, Jungbunzlauer Ladenburg GmbH; Email: teresa.berninger@jungbunzlauer.com
References

Table 1: Comparison of different thickeners in natural, sulfate-free formulation
Xanthan gum is a polymer of repeating sugar units somewhat similar to cellulose. But unlike cellulose, xanthan gum possesses a trisaccharide moiety present along the backbone of the molecule, repeating every other glycosyl residue. Thanks to its distinctive structure, xanthan gum provides a host of benefits when used as a thickener. It provides excellent thickening even at low concentrations. It is resilient to pH variations; therefore, it is stable in both alkaline and acidic conditions. It also possesses very good thermal stability so it can be easily added into hot or cold processes and it is compatible with a wide variety of other ingredients. In shampoos, it exhibits very good compatibility with surfactants and has shown positive effects on foam formation, height and stability.2
When choosing surfactants, one must consider the move toward sulfate-free varieties to create milder formulations. In the context of natural ingredients, there also exist a number of bio-based options with great potential. With any new class of ingredients come challenges in how they may impact performance or interact with other raw materials. For example, traditional thickening of formulations by adding salt is not feasible with some bio-based surfactants. The aim of this study was to see if the unique qualities of xanthan gum could benefit formulators looking to utilize these surfactants. One consideration with xanthan gum is that standard grades may not be able to replace traditional thickeners without compromising the appearance, texture or feel of the product. By tweaking the processing, unique grades of xanthan gum with reduced pseudoplasticity were developed to alleviate these potential sensory issues. We will explore such a grade by highlighting its utility in a sulfate-free shampoo.
Results & Discussions
In order to better understand the behavior of a reduced pseudoplastic xanthan gum with surfactants, a variety of nonionic, anionic and amphoteric surfactants were dissolved into a 1% w/w xanthan gum solution at varying concentrations. After dissolution, the pH was adjusted to 5.5, and the resulting samples were monitored for one month. During this time, optical evaluations and viscosity measurements were made. Glycolipids, coco-glucosides, caprylol/caproyl methyl glucamide, sodium cocoyl glutamate and cocamidopropyl betaine were among the range of sulfate-free, bio-based surfactants tested. Since it is rare that only single surfactant systems are used in formulations, dual-combinations of the surfactants were also dissolved into 1% xanthan gum solutions. An active concentration of 12% was chosen for each surfactant combination. Further, a 1:1 ratio was selected when it was not possible to categorize main- and co-surfactants. If categorization was possible, a ratio of 5:1 was tested. All of the surfactants and surfactant blends exhibited excellent stability in combination with xanthan gum.

To measure the flow curve at 21°C, a Haake Rheostress rheometer with cone-plate geometry (C35 / 2°Ti L) was used. The apparent viscosity was measured by an increasing shear rate from 0.01 to 100 s-1. The storage and the behavior on the hand after application are described by a low shear rate. A medium shear rate represents the application on the hair. Squeezing the shampoo out of the bottle is represented by a high shear rate.3,4
As a first step, different benchmarks were measured to define representative target values of viscosity (Fig. 1). Depending on the thickener, different flow behaviors were observed. The benchmark, thickened with a combination of salt and PEG, showed Newtonian behavior, which means with an increasing shear rate the apparent viscosity remained constant. The apparent viscosity of the benchmark thickened with xanthan gum decreased with an increasing shear rate, so the benchmark containing xanthan gum showed shear-thinning behavior.5
To understand the rheological behavior of different thickeners with natural surfactants, thickeners were combined with a typical, natural surfactant system. This system included a combination of sodium cocoyl glutamate, cocamidopropyl betaine and coco glucoside in a 2:2:1 ratio based on active substance. The total concentration of this system was 12% w/w, also based on active substance. The aim was to come as close to the apparent viscosity defined by the benchmarks

Figure 1: Rheology results
Thickening via salt addition was the first approach evaluated since it is the traditional way of increasing viscosity. A hydrophobic thickener (cocamide DEA), a hydrophilic thickener (PEG-150 distearate) and a synthetic gelling agent (acrylates/C10-13 alkyl acrylate crosspolymer) were also tested to compare the rheological behavior to a surfactant system thickened with xanthan gum. As mentioned previously, there are multiple grades of xanthan gum each with different flow behaviors. For this study we looked at two clear solution grades, the first one with a standard flow behavior (FNCS-PC) and the second one with a reduced pseudoplasticity (FNCSP-PC). The amount of thickening agent was adapted to the rheological target values from the benchmark. The formulation is shown in Table 1.
The flow curves are shown in Fig. 1. The variability in flow behavior results from different thickening mechanisms. Salt and cocamide DEA are promoting the arrangement of surfactants into rod-shaped micelles, forming a thickening network with Newtonian behavior. Also the surfactant system thickened with PEG-150 distearate exhibits Newtonian flow behavior due to the crosslinking of the micelles and the increase of their size. In contrast, xanthan gum and the synthetic gelling agent (acrylates/C10-13 alkyl acrylate crosspolymer) are thickening the aqueous phase, which leads to shear thinning behavior of the thickened surfactant system.6
Our model system of natural and sulfate-free surfactants could not be thickened with salt or cocamide DEA. The viscosity did not reach the defined target even with elevated concentrations of salt which highlights the problem formulators face trying to thicken natural, sulfate-free surfactant systems. The traditional approach of thickening with salt does not work. The desired viscosity can be reached by using PEG-150 distearate; however, PEG derivatives may be undesirable from a consumer point of view, since they facilitate penetration of potentially harmful substances through the skin.7 Xanthan gum and the synthetic gelling agent (acrylates/C10-13 alkyl acrylate crosspolymer) show the desired viscosity. However, as awareness for synthetic polymers rises, the synthetic gelling agent (acrylates/C10-13 alkyl acrylate crosspolymer) may be viewed by the consumers as “microplastic.”1

Table 2. Xanthan gum thickened sulfate-free shampoo (pH adjusted to 5.2-5.7).
Discussion of Sensory Data
Sensory testing was performed to see just how close a reduced pseudoplastic xanthan gum could come to feeling like a traditionally thickened formulation. A group of 12 trained panelists was asked to compare benchmark, commercially available sulfate-free shampoo thickened with PEG and salt to a sulfate-free formulation successfully thickened with xanthan gum FNCSP-PC. Table 2 describes the test formulation in detail.
The panelists were given two shampoos and asked to compare the controllability of dosing, viscosity and haptics. In these three particular categories, a preference was indicated. Based on the results shown in Fig. 2, there was not a significant difference in the preference between the benchmark shampoo and the xanthan gum thickened sulfate-free formulation. Thus, we can conclude that xanthan gum FNCSP-PC

Figure 2. Results of the sensory evaluation.
Conclusion
While most thickeners may work just fine in combination with surfactants like SLS and SLES in shampoo formulations, new regulations and the move toward more bio-based surfactants may pose significant challenges. Based off the stability studies, rheological measurements, and sensory evaluation, it is clear that one can effectively thicken in the presence of bio-based, sulfate-free surfactants without compromising the feel of the product using xanthan gum. In addition, xanthan gum is appropriate for natural formulations. This data demonstrates that xanthan gum is a powerful tool for formulators looking to clean up their labels.
More information
Amirah Bajawi, application technology assistant, Jungbunzlauer Ladenburg GmbH, Email: amirah.bajawi@jungbunzlauer.com; Cameron Whitney, market development manager, Jungbunzlauer Inc.; Email: cameron.whitney@jungbunzlauer.com; Dr. Teresa Berningers, senior project manager application technology, Jungbunzlauer Ladenburg GmbH; Email: teresa.berninger@jungbunzlauer.com
References
- E. C. Agency, “ANNEX to the ANNEX XV RESTRICTION REPORT – intentionally added microplastics. ECHA Propos. A Restrict. 1.2,” pp. 139–181, 2019.
- G. Marin Velasquez, M. Neubauer, T. Willers, and V. Vill, “The Influence of Natural Gelling Agents on the Foaming Behavior and Foam Structure in Surfactant Systems,” SOFW-Journal, vol. 1/2-2020, pp. 20-25, 2020.
- P. A. Cornwell, “A review of shampoo surfactant technology: Consumer benefits, raw materials and recent developments,” Int. J. Cosmet. Sci., vol. 40, pp. 16–30, 2017, doi: 10.1111/ics.12439.
- C. Savary, G., Grisel, M. and Picard, “Cosmetics and Personal Care Products,” in Natural Polymers, O. Olatunji, Ed. Springer, 2016, pp. 219–261.
- T. G. Mezger, Das Rheologie Handbuch: Für Anwender von Rotations- und Oszillations-Rheometern. Vincentz Network GmbH & Co. KG, 2006.
- U. Kortemeier, J. Venzmer, A. Howe, B. Grüning, and S. Herrwerth, “Thickening Agents for Surfactant Systems,” SOFW-Journal, vol. 136, pp. 29–38, 2010.
- H.-J. Jang, C. Y. Shin, and K.-B. Kim, “Safety Evaluation of Polyethylene Glycol (PEG) Compounds for Cosmetic Use Hyun-Jun,” Toxicol. Res., vol. 31, no. 2, pp. 173–180, 2015, doi: 10.5487/TR.2015.31.2.173.














