Paolo Giacomoni, PhD, Insight Analysis Consulting11.01.21
Despite the creation of words like cosmeceutical and quasi-drugs, we maintain firm in our mind that cosmetics and skin care products only are legally allowed to affect the surface. Claims of biochemical efficacy, let alone of physiological efficacy, will condemn the products into the drug category so that their development costs will become prohibitive. The self-censorship that ensues makes it so that brands prefer to advertise their products as “snake oils,” insisting on buzzwords and concealing any possible underlying mechanism of their efficacy.
This situation is, of course, grotesque. It is the perverse consequence of what was the reasonable assumption made over one century ago, that the physical modification of the surface of the skin does not entail physiological modifications of the surface itself. We do now know that adding molecules to the surface of the skin (water, vitamins, antioxidants, lipids, botanical extracts) interferes with dozens of biochemical pathways affecting the physiology of the epidermis. Even the one and only cosmetic activity for which an efficacy claim is accepted, (i.e. exfoliation), results at least in stimulating nerve cells (stinging), activating cellular duplication and altering the microbiome.
While we wait for the US Food and Drug Administration (FDA) guidelines for skin care products to be inspired by reason, we can add to our testing panoply a test that will justify the efficacy of our products while respecting the FDA mandate—that cosmetic products can only modify the surface.
Assessing Surface Modification
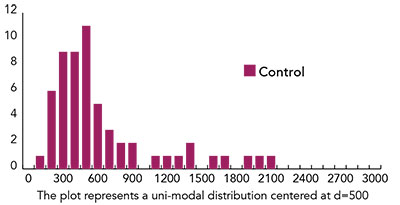
Fig. 2: Histogram of distribution of d-values for 58 untreated controls.
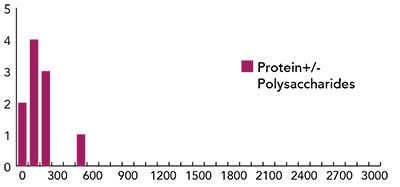
Fig. 3: Histogram of distribution of d-values for specimens treated with ten protein-polysaccharide mixtures.
The skin is covered by the stratum corneum (aka horny layer) that is made of squames. The horny layer can be rough and lack smoothness, and can therefore scatter the incident light, enhancing the visibility of fine lines, wrinkles, age spots and other surface imperfections. Testing surface modification in vivo is costly: it requires large cohorts of volunteers as well as trained expert panels for clinical analysis. In addition, the instrumental determination of the exfoliating effects of a product is only feasible by measuring the increase of the transepidermal water loss (TEWL). Unfortunately, TEWL is a parameter too often illegitimately used to assess the integrity of the “barrier.” Low TEWL would correspond to intact barrier, so that any increase of TEWL would be interpreted as barrier disruption and would therefore be hardly suitable to convey the message of safe and effective modification of the surface.
These difficulties can be circumvented with the use of reconstructed skin. Reconstructed skin is made by seeding cultured human keratinocytes on the top of an equivalent dermis. The equivalent dermis is a collagen gel containing human fibroblasts. The keratinocytes grow, differentiate and produce an observable stratum corneum, similar to the one naturally occurring in vivo.
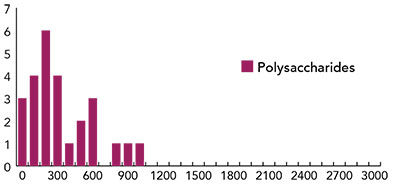
Fig. 4: Histogram of distribution of d-values for specimens treated with 26 polysaccharide.
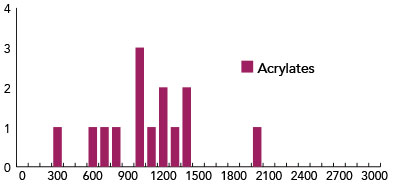
Fig. 5: Histogram of distribution of d-values for 14 specimens treated with acrylates or polyacrylates.
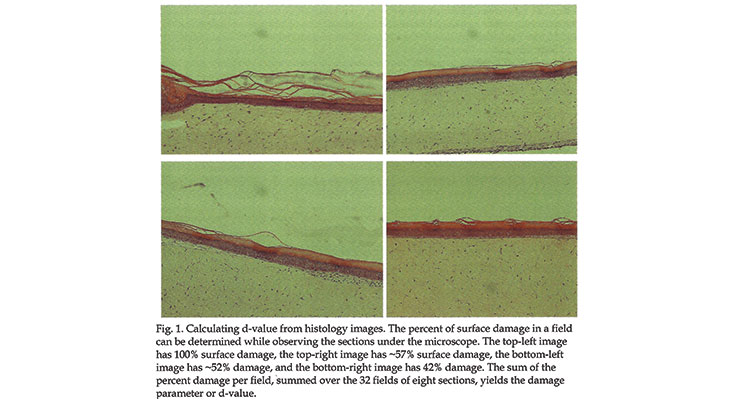
One can assess the damage “d” of the surface by taking “biopsies” and preparing histology sections from samples of reconstructed skin, untreated or treated with the ingredient of interest, as follows.1
One biopsy per treated sample. Prepare several histology sections per biopsy.
Analyze eight histology sections per biopsy (four fields per section) for a total of 32 fields.
In every microscopy field, one can visually assess what percentage of the stratum corneum is detached.
Summing the % of “damaged” surface in every field, the condition of the surface is defined by a number “d” such that 0 < d < 3200.
An example of this assessment is given in Figure 1: (top left: d= 100%; top right: d=57%; bottom left: d=52% and bottom right: d= 42%)
Since it is impossible to assess the status of the stratum corneum before and after the treatment on the same sample of reconstructed skin, one must assess the status of the surface on a large number of untreated samples and then treat other samples with the ingredient of interest. The comparison of the status of the treated sample with the status of the control samples will reveal the efficacy of the tested ingredient.
An example of the assessment of the status of the surface is given in Figure 2.
From the results in Figure 2 one learns that 45 out of 58 untreated samples have a value of the “d” parameter between 300 and 900. If we assume this to be the case for all the samples of reconstructed skin in our hands, we can suggest that a treatment resulting in a “d” value less than 300 be considered as a treatment that repairs the surface, whereas a treatment that results in a “d” value larger than 900 can be considered to be a treatment that damages the surface.
Samples were treated with different ingredients and the status of the surface are reported in the figures above right.
Figure 3 displays the results for samples treated with mixtures of proteins and polysaccharides. Figure 4 reports the status of surface treated with polysaccharides only, and Figure 5 shows the effects of treating reconstructed skin with acrylates of poly-acrylates. From the analysis of the figures it appears that the acrylates and poly-acrylates used in this experiment cannot be said to improve the status of the surface, whereas mixtures of proteins and polysaccharides, by resulting in “d” values smaller than 300, can be said to have the effect of repairing the surface. On the other hand, different polysaccharides appear to have different surface effects.
Conclusion
The method described is a quantitative method to assess the surface repairing effect of ingredients of cosmetic interest. It is easy to perform and is cost effective. It allows one to assess the capability of ingredients to modify the surface without implying physiological changes. It could become a test of choice for assessing cosmetic activity, and should be recommended by the FDA.
Am I dreaming?
References
Paolo Giacomoni, PhD
Insight Analysis Consulting
[email protected]
516-769-6904
Paolo Giacomoni acts as an independent consultant to the skin care industry. He served as executive director of research at Estée Lauder and was head of the department of biology with L’Oréal. He has built a record of achievements through research on DNA damage and metabolic impairment induced by UV radiation as well as on the positive effects of vitamins and antioxidants. He has authored more than 100 peer-reviewed publications and has more than 20 patents.
This situation is, of course, grotesque. It is the perverse consequence of what was the reasonable assumption made over one century ago, that the physical modification of the surface of the skin does not entail physiological modifications of the surface itself. We do now know that adding molecules to the surface of the skin (water, vitamins, antioxidants, lipids, botanical extracts) interferes with dozens of biochemical pathways affecting the physiology of the epidermis. Even the one and only cosmetic activity for which an efficacy claim is accepted, (i.e. exfoliation), results at least in stimulating nerve cells (stinging), activating cellular duplication and altering the microbiome.
While we wait for the US Food and Drug Administration (FDA) guidelines for skin care products to be inspired by reason, we can add to our testing panoply a test that will justify the efficacy of our products while respecting the FDA mandate—that cosmetic products can only modify the surface.
Assessing Surface Modification

Fig. 2: Histogram of distribution of d-values for 58 untreated controls.

Fig. 3: Histogram of distribution of d-values for specimens treated with ten protein-polysaccharide mixtures.
These difficulties can be circumvented with the use of reconstructed skin. Reconstructed skin is made by seeding cultured human keratinocytes on the top of an equivalent dermis. The equivalent dermis is a collagen gel containing human fibroblasts. The keratinocytes grow, differentiate and produce an observable stratum corneum, similar to the one naturally occurring in vivo.

Fig. 4: Histogram of distribution of d-values for specimens treated with 26 polysaccharide.

Fig. 5: Histogram of distribution of d-values for 14 specimens treated with acrylates or polyacrylates.
One biopsy per treated sample. Prepare several histology sections per biopsy.
Analyze eight histology sections per biopsy (four fields per section) for a total of 32 fields.
In every microscopy field, one can visually assess what percentage of the stratum corneum is detached.
Summing the % of “damaged” surface in every field, the condition of the surface is defined by a number “d” such that 0 < d < 3200.
An example of this assessment is given in Figure 1: (top left: d= 100%; top right: d=57%; bottom left: d=52% and bottom right: d= 42%)
Since it is impossible to assess the status of the stratum corneum before and after the treatment on the same sample of reconstructed skin, one must assess the status of the surface on a large number of untreated samples and then treat other samples with the ingredient of interest. The comparison of the status of the treated sample with the status of the control samples will reveal the efficacy of the tested ingredient.
An example of the assessment of the status of the surface is given in Figure 2.
From the results in Figure 2 one learns that 45 out of 58 untreated samples have a value of the “d” parameter between 300 and 900. If we assume this to be the case for all the samples of reconstructed skin in our hands, we can suggest that a treatment resulting in a “d” value less than 300 be considered as a treatment that repairs the surface, whereas a treatment that results in a “d” value larger than 900 can be considered to be a treatment that damages the surface.
Samples were treated with different ingredients and the status of the surface are reported in the figures above right.
Figure 3 displays the results for samples treated with mixtures of proteins and polysaccharides. Figure 4 reports the status of surface treated with polysaccharides only, and Figure 5 shows the effects of treating reconstructed skin with acrylates of poly-acrylates. From the analysis of the figures it appears that the acrylates and poly-acrylates used in this experiment cannot be said to improve the status of the surface, whereas mixtures of proteins and polysaccharides, by resulting in “d” values smaller than 300, can be said to have the effect of repairing the surface. On the other hand, different polysaccharides appear to have different surface effects.
Conclusion
The method described is a quantitative method to assess the surface repairing effect of ingredients of cosmetic interest. It is easy to perform and is cost effective. It allows one to assess the capability of ingredients to modify the surface without implying physiological changes. It could become a test of choice for assessing cosmetic activity, and should be recommended by the FDA.
Am I dreaming?
References
- Giacomoni, PU (2011) Use of Reconstructed Skin Specimens to Analyze Stratum corneum Remodeling and Epidermal Modifications in: Skin Biopsy - Perspectives (Uday Kopkar Editor) Intech (Rijeka, Croatia) pages 225-240 Open Access
Paolo Giacomoni, PhD
Insight Analysis Consulting
[email protected]
516-769-6904
Paolo Giacomoni acts as an independent consultant to the skin care industry. He served as executive director of research at Estée Lauder and was head of the department of biology with L’Oréal. He has built a record of achievements through research on DNA damage and metabolic impairment induced by UV radiation as well as on the positive effects of vitamins and antioxidants. He has authored more than 100 peer-reviewed publications and has more than 20 patents.