01.31.23
According to a December 2022 Warning Letter, the US Food & Drug Administration (FDA) has concerns about the lab that tested and found benzene in a number of aerosol sunscreens and other products.
The letter sent to Valisure CEO David Y.Y. Light is dated Dec. 5, 2022. In the letter, FDA said its inspectors found “methodological deficiencies” that included Valisure’s failure “to establish and document the accuracy, sensitivity, specificity and reproducibility of test methods.”
FDA also noted concerns about GMP and analytical discrepancies in Valisure’s testing methods.
Valisure, which is based in New Haven, CT, expanded its testing capabilities to include analysis for benzene in late 2020. At the same time, it broadened the type of products it analyzed for pharmacy partners to include over-the-counter items and consumer healthcare products. Valisure sold its pharmacy subsidiary in 2021.
Source: Valisure
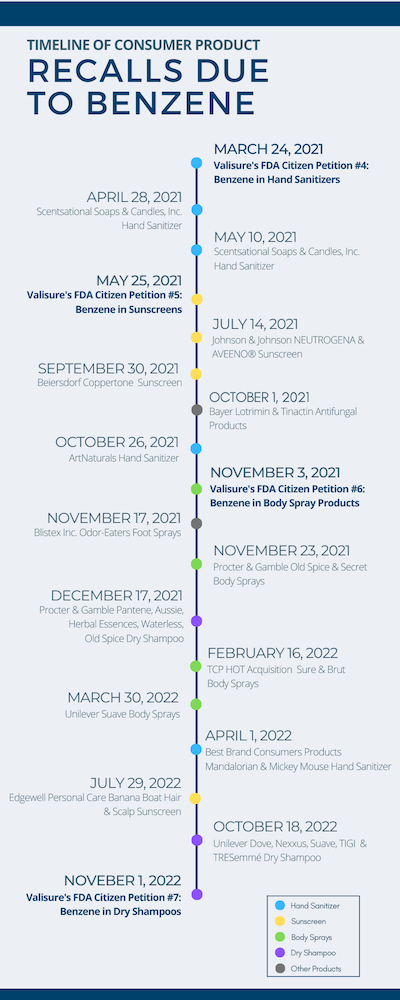
Since 2021, Valisure has issued seven FDA Citizen Petitions about benzene in consumer products, including sunscreen sprays, body sprays, foots sprays, hand sanitizers and, most recently, dry shampoos. On Nov. 1, 2022, the company said it detected high levels of benzene in several brands and batches of dry shampoo products.
In March 2021, Valisure filed its Citizen Petition on benzene contamination in hand sanitizer. In May 2021, it filed a Citizen Petition on benzene contamination in sunscreens. In November 2021 it filed a Citizen Petition on benzene contamination in body sprays (see image)
The reports filed by Valisure led to high profile recalls of personal care products.
According to Aboutlawsuits.com, the testing laboratory has “drawn the ire of pharmaceutical companies, manufacturers and federal regulators.”
Experts in the personal care industry have issued opinions as to why trace amount of benzene had been found in some sprays; some noted that supply chain issues during the pandemic could be the cause.
Just last week, Edgewell expanded its recall product list beyond sunscreens after hair and scalp sprays were found to have trace amounts of benzene.
On Dec. 6, 2022, Florida Judge Robin L. Rosenberg dismissed the Zantac multi-district litigation (MDL) case. In the judge’s opinion, the plaintiff’s expert failed to use reliable methods to connect Zantac with cancer, according to a post by Forbes.
Large brands in topical personal care, including those owned by Procter & Gamble, Johnson & Johnson, Unilever, Edgewell and others, have pulled products from shelves; they also settled or continue to face lawsuits.
The letter sent to Valisure CEO David Y.Y. Light is dated Dec. 5, 2022. In the letter, FDA said its inspectors found “methodological deficiencies” that included Valisure’s failure “to establish and document the accuracy, sensitivity, specificity and reproducibility of test methods.”
FDA also noted concerns about GMP and analytical discrepancies in Valisure’s testing methods.
Valisure, which is based in New Haven, CT, expanded its testing capabilities to include analysis for benzene in late 2020. At the same time, it broadened the type of products it analyzed for pharmacy partners to include over-the-counter items and consumer healthcare products. Valisure sold its pharmacy subsidiary in 2021.

Source: Valisure
In March 2021, Valisure filed its Citizen Petition on benzene contamination in hand sanitizer. In May 2021, it filed a Citizen Petition on benzene contamination in sunscreens. In November 2021 it filed a Citizen Petition on benzene contamination in body sprays (see image)
The reports filed by Valisure led to high profile recalls of personal care products.
According to Aboutlawsuits.com, the testing laboratory has “drawn the ire of pharmaceutical companies, manufacturers and federal regulators.”
Experts in the personal care industry have issued opinions as to why trace amount of benzene had been found in some sprays; some noted that supply chain issues during the pandemic could be the cause.
Just last week, Edgewell expanded its recall product list beyond sunscreens after hair and scalp sprays were found to have trace amounts of benzene.
Zantac Case Withdrawn
Valisure gained national attention when its testing reportedly found high levels of NDMA in heartburn drug Zantac 2019. Zantac was pulled from store shelves, and a class lawsuit followed.On Dec. 6, 2022, Florida Judge Robin L. Rosenberg dismissed the Zantac multi-district litigation (MDL) case. In the judge’s opinion, the plaintiff’s expert failed to use reliable methods to connect Zantac with cancer, according to a post by Forbes.
Large brands in topical personal care, including those owned by Procter & Gamble, Johnson & Johnson, Unilever, Edgewell and others, have pulled products from shelves; they also settled or continue to face lawsuits.












