Christine Esposito, Managing Editor02.17.21
Evolved Ayurvedic Discoveries, Inc./BioCBDPlus has been sent a warning letter by the Food and Drug Administration (FDA) following a review of its website.
According to FDA, the Encinitas, CA-based company offers CBD products that are intended to mitigate, prevent, treat, diagnose, or cure COVID-191 in people. The claims on the company’s website and social media websites establish that its products—including BioCBD+ Total Body Care and BioCBD+ Topical Oil Muscle & Joint Support—are unapproved new drugs sold in violation of sections 505(a) and 301(d) of the Federal Food, Drug, and Cosmetic Act. In addition, FDA said the products are misbranded drugs sold in violation of section 502(f)(1) of the FD&C Act.
The review was conducted from August through October 2020.
According to FDA’s letter, from September 18, 2020 posts to the company’s Facebook and Instagram pages included:
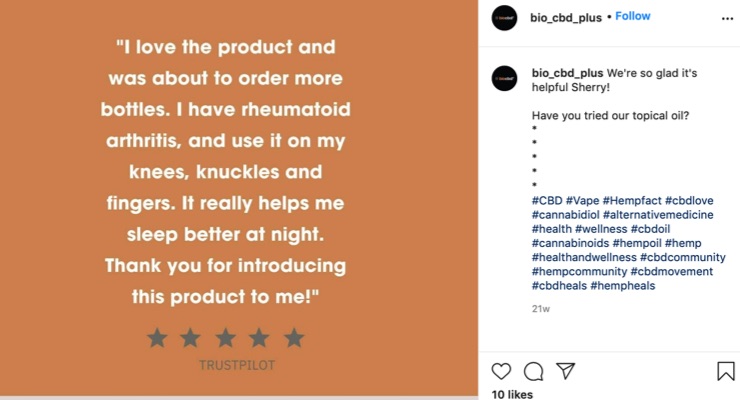
“Have you tried our topical oil?” accompanied by a graphic with the text ‘I love the product and was about to order more bottles. I have rheumatoid arthritis, and use it on my knees, knuckles and fingers. It really helps me sleep better at night.’”
In addition, a February 26, 2020Instagram post stated:
“‘BioCBD+ has been a huge relief to my quality of life with Crohns, anxiety . . . #cbdoil’ accompanied by a graphic displaying “BioCBD+ Total Body Care” and “BioCBD+ Topical Oil Muscle & Joint Support.”
FDA said that the company’s BioCBD+ products are not generally recognized as safe and effective for their above referenced uses and, therefore, these products are “new drugs." The agency also said the products were misbranded in that their labeling fails to bear adequate directions for use.
The agency also highlighted the company’s BioCBD+ Peace Vape product as “particularly concerning” because the ingredients and potential impurities in oral inhalation products may trigger laryngospasm and bronchospasm and may be toxic to the tissues in the upper or lower airways.
According to FDA, the Encinitas, CA-based company offers CBD products that are intended to mitigate, prevent, treat, diagnose, or cure COVID-191 in people. The claims on the company’s website and social media websites establish that its products—including BioCBD+ Total Body Care and BioCBD+ Topical Oil Muscle & Joint Support—are unapproved new drugs sold in violation of sections 505(a) and 301(d) of the Federal Food, Drug, and Cosmetic Act. In addition, FDA said the products are misbranded drugs sold in violation of section 502(f)(1) of the FD&C Act.
The review was conducted from August through October 2020.
According to FDA’s letter, from September 18, 2020 posts to the company’s Facebook and Instagram pages included:
“Have you tried our topical oil?” accompanied by a graphic with the text ‘I love the product and was about to order more bottles. I have rheumatoid arthritis, and use it on my knees, knuckles and fingers. It really helps me sleep better at night.’”
In addition, a February 26, 2020Instagram post stated:
“‘BioCBD+ has been a huge relief to my quality of life with Crohns, anxiety . . . #cbdoil’ accompanied by a graphic displaying “BioCBD+ Total Body Care” and “BioCBD+ Topical Oil Muscle & Joint Support.”
FDA said that the company’s BioCBD+ products are not generally recognized as safe and effective for their above referenced uses and, therefore, these products are “new drugs." The agency also said the products were misbranded in that their labeling fails to bear adequate directions for use.
The agency also highlighted the company’s BioCBD+ Peace Vape product as “particularly concerning” because the ingredients and potential impurities in oral inhalation products may trigger laryngospasm and bronchospasm and may be toxic to the tissues in the upper or lower airways.