11.22.23
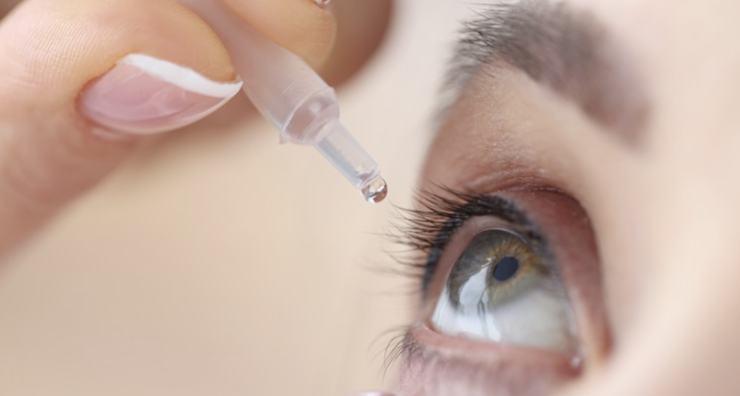
Kilitch Healthcare India Limited is recalling eyedrops with expiration dates ranging from November 2023 to September 2025. The move comes after Food and Drug Administration investigators found insanitary conditions at the place of manufacture, posing a potential risk of eye infections or related harm. FDA is warning consumers not to purchase and to immediately stop using 26 over-the-counter eye drop products due to the potential risk of eye infections that could result in partial vision loss or blindness. The recalled products were sold nationwide by retailers including CVS, Rite Aid and Walmart.
This announcement comes weeks after FDA warned consumers not to purchase or use certain methylsulfonylmethane (MSM) eye drops due to contamination.
The recall is just the latest in a series of contaminatiion issues regarding eyedrops. Of course, contamination isn't limited to eye drops. That's why the Personal Care Products Council and the Quality Committee Manufacturing Hygiene Task Force teamed with the University of Cincinnati College of Pharmacy to create a post graduate certificate course devoted to manufacturing hygiene.
J&J Recalls 8,300 Bottles of Clean & Clear Cleanser for Micro Contamination
Preservative Solutions Ensure Safety for Beauty Consumers
This announcement comes weeks after FDA warned consumers not to purchase or use certain methylsulfonylmethane (MSM) eye drops due to contamination.
The recall is just the latest in a series of contaminatiion issues regarding eyedrops. Of course, contamination isn't limited to eye drops. That's why the Personal Care Products Council and the Quality Committee Manufacturing Hygiene Task Force teamed with the University of Cincinnati College of Pharmacy to create a post graduate certificate course devoted to manufacturing hygiene.
Related News
Preservative-Free Eye Drops Linked to Bacterial Infection and DeathJ&J Recalls 8,300 Bottles of Clean & Clear Cleanser for Micro Contamination
Preservative Solutions Ensure Safety for Beauty Consumers