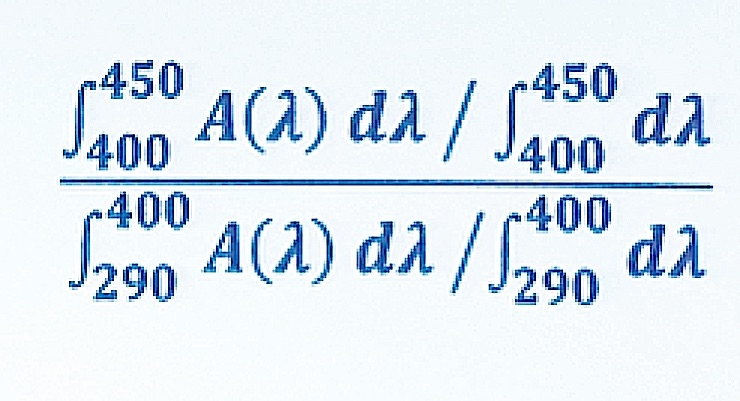10.12.18
Mingling cosmetics and pharmaceuticals may be frowned upon by the US Food and Drug Administration, but professionals from both fields were on a level playing field at this week’s Innovations in Dermatological Sciences Conference in Iselin, NJ. The event was organized by Dr. Bozena “Bo” Michniak-Kohn, founder and director of the Center for Dermal Research (CDR) and Professor of Pharmaceutics at Rutgers-The State University of New Jersey. The Innovations in Dermatological Sciences conference was the sixth in the series of annual events put on by CDR.
The theme for this year’s conference was “Future of Dermatologicals and Cosmeceuticals.” The meeting was attended by more than 180 participants and over 20 sponsors and donors. This was the highest attendance of any of the previous meetings, according to Michniak-Kohn. Companies represented included Rodan + Fields, Johnson & Johnson, MedPharm, Logan Instruments Corp., Tergus Pharma, Dow Development, Gattefosse, Silab, BASF, Jeen International, Galderma-Nestle Skin Health, GenoSkin, Evonik, Genemarkers, Beri Esthetique, Colgate-Palmolive, L’Oréal, Lubrizol, Novan, Avon Products, PCA Skin and Tioga Research.
There were 21 speakers and a regulatory panel which included a speaker from the FDA, Dr. Nakissa Sadrieh, director of the agency’s cosmetics division. In addition, the poster session attracted more than 20 posters in the skin research areas. Attendees came from all over the US as well as internationally (primarily Europe and South America), according to Michniak-Kohn. The annual award of CDR Sponsor of the Year was awarded to Tergus Pharma.
Startling Statistics
Leading off was a keynote by Dr. David Margolis of the University of Pennsylvania, who noted that in 2013, 85 million Americans, 25% of the population, saw a dermatologist and were treated for 1.6 skin diseases. That same year, $75 billion was spent on skin care treatment; $46 billion was attributable to medical costs (office visits, procedure and test), $15 billion to prescription drugs and $4 billion to vaccines (58%), other skin procedures (42%) and skin cancer screening (0.1%), which corresponds to 3.8% of 2013 total US health care costs. The cost per capita was $240; for comparison, diabetes is $950; cardiovascular disease is $950 and renal failure is $147.
Cost corresponding to OTC products for skin disease was nearly $10 billion. “For OTC, that works out $32 per year per person for skin diseases,” noted Margolis. “Most of the drugs used by dermatologists were not originally implicated for skin diseases.”
On a global scale, skin disease is a leading cause of disease burden. Margolis pointed to a 2014 study in the Journal of Investigative Dermatology which found that three skin conditions—fungal skin diseases, other skin and subcutaneous diseases, and acne—were in the top 10 most prevalent diseases worldwide in 2010, and eight fell into the top 50; these additional five skin problems were pruritus, eczema, impetigo, scabies and molluscum contagiosum. Yet, very few studies, according to Margolis, have directly focused on skin disease.
He reminded the audience that the US and global populations are getting older, which means a growing demand for products to treat chronic wounds such as diabetic foot ulcers, which is the No. 1 cause of amputations in the US. In 1980, there were 5.5 million cases of diabetes; by 2014, the number of cases had climbed to 24 million, according to Margolis, who also noted that people who have amputations are 20% more likely to die in any given year.
Psoriasis and atopic dermatitis also demand attention from doctors and formulators. Margolis noted that psoriasis affects 2-4% of the adult population and is associated with an aging population. In contrast, atopic dermatitis is prevalent in 10-20% of children in industrialized nations and is associated with other atopic illnesses such as seasonal allergies, asthma, and drug and food allergies. No wonder why so many companies are focused on psoriasis and atopic dermatitis.
Novan president Nathan Stasko explained how a 100% nitric oxide treatment developed by his company effectively treats inflammation, acne, atopic dermatitis and psoriasis. The company’s Nitricil technology enables the delivery of nitric oxide by storing the gaseous species on large polymers, which has led to the development of a diverse pipeline of time-release, nitric oxide-releasing new chemical entities for short- or long-term healthcare applications. The result, according to Novan, is a stabilized, drug-able nitric oxide that can be formulated into gels, ointments and creams.
There are a range of dermal penetration enhancers that may be incorporated into topical pharmaceutical formulations, noted Inayet Ellis of Gattefossé; these include hydroalcoholic solvents (alcohols, ether alcohols, glycols); lipophilic solvents (esters, fatty esters) and surfactants (nonionic, anionic and cationic).
“Hydration of the stratum corneum promotes absorption of both hydrophilic and lipophilic materials. Proper use of excipients and formulation design facilitate passage across the stratum corneum and drive diffusion past the stratum corneum,” concluded Ellis. “Safe delivery of medicines via the skin is possible, but the formulator must strike the right balance between solubilization, partitioning and localization.”
A Rise in Pollutants
According to the World Health Organization (WHO), more than 90% of the urban population live with pollutant levels above the WHO-defined standard limits. Yong Zhuang of Avon Products reviewed some of the most common pollutants including polycyclic aromatic hydrocarbons (PAHs), which he called among the most widespread and dangerous organic pollutants. PAHs are converted into quinines, redox-cycling chemicals that produce reactive oxygen species (ROS), which in turn, damage DNA and lead to skin aging. Some of the environmental aggressors that produce oxidative damage to skin include hydrogen peroxide, UVA/B and urban dust. However, Avon researchers identified a blend of antioxidants that can help prevent and/or repair damage caused by a number of environmental aggressors in vitro. Finally, a topical formula (Anew Skinvincible) incorporating this blend of antioxidants has been shown to improve skin health in a clinical study.
Yarka Kepic, Verdi Enterprises Inc., reviewed how efficacy levels of bioactive neuropeptides are impacting neurocosmetic formulations. She noted that these materials are effective at 5 parts per million compared to 700 parts per million for retinol. Other neurocosmetic actives include cinnamaldehyde, monks pepper, pumpkin seed extract and dihydroavenanthramide D. In the not-to-distant-future, these and other actives will improve the efficacy of a range of cosmetic and OTC products including premature aging, sunless tanning, chronic pain and itch, wound healing, acne rosacea and psoriasis, she explained.
“Future dermaceutical and cosmeceutical products will rely on increased use of synthetic biometric peptides,” she concluded. “There will be increased use of sustainable materials which elicit a neurogenic response.”
Retinoids increase fibroblast growth and dermal collagen synthesis, while decreasing levels of MMPs, according to PCA Skin’s Ivana Veljkovic who reviewed the material’s history and mechanism of action in skin formulations.
“Retinoids have been implicated in nearly all phases of epidermal differentiation and keratinization,” she added.
The Role of Retinoids
Veljkovic admitted that initially, acne therapy with retinoids can be frustrating, as in the first few weeks of use, the follicular epithelium loosens and, when mixed with pro-inflammatory lipids in sebum, can lead to increased breakouts. To reduce this negative effect, retinoid are often used in tandem with oral and topical antibacterials, such as benzoyl peroxide. Despite this drawback, retinoids remain the most effective comedolytic agents making them appropriate for comedonal and inflammatory acne treatments, according to Veljkovic. Now, retinoids are thought to have an effect on intrinsic aging.
“This is important, as previously, it was believed that we could only impact extrinsic aging and all intrinsic aging was inevitable,” she explained.
With regular use of retinoids, smoother skin usually occurs in the first four weeks and is likely the result of an increase in epidermal proliferation and consequential epidermal thickening, compaction of the stratum corneum and deposition of glycosaminoglycans in the stratum corneum and in the intercellular spaces of the epidermis. Retinoids reduce pigment levels in skin by inhibiting melanogensis, inhibiting the transfer of melanin from melanocytes to keratinocytes, and increasing the shedding of melanin-laden keratinocytes as a result of epidermal proliferation, explained Veljkovic.
“Despite the benefits of topical retinoids, instability makes it extremely hard to use them effectively in formulations,” she noted.
At the same time, retinoids can be very irritating. To overcome some of these limitations, PCA relies on non-irritating molecules (OmniSomes) to keep retinol protected and stable and increase skin penetration. Another PCA strategy is use of the pro-drug concept, whereby retinol is well-delivered and converted into the biologically active form, retinoic acid, with reduced irritancy. Of course, each formula includes ancillary ingredients to enhance each product’s overall efficacy, she concluded.
PCA has also maximized L-ascorbic acid-based skin care with the launch of C&E Advanced with Hexylresorcinol and Silymarin. According to PCA the formula provides “unparalleled” antioxidant protection by combining four antioxidants that minimize the appearance of lines and wrinkles, reduces hyperpigmentation, protects skin from environmental stressors and strengthens and smooths skin. The formula contains 20% L-ascorbic acid, 5% tocopherol, 1% silymarin and 1% hexylresorcinol.
“(Our) anhydrous delivery technology allows for maximum stabilization of L-ascorbic acid over extended periods of time,” said Veljkovic. “The unique transdermal delivery system allows for full delivery of water-soluble, L-ascorbic acid into the skin over two hours.”
Feeling Blue
Olga Dueva-Koganov, Rodan + Fields, reviewed the latest trends in solar irradiation protection, noting that consumers are becoming increasingly aware of the negative impact of irradiation on the skin beyond UV; e.g., blue light (HEV). In one survey, 28% of Brazilians said they would like to try sun care products that offer protection from visible light. However, existing methods do not explicitly assess the HEV light region.
“To enable the evaluation of sunscreen’s protection potential in the blue light region, we modified Diffy’s equation and utilized it for the evaluation of commercial sunscreen products,” explained Dueva-Koganov. (The modified equation can be found in the slide show in this article.)
Rodan + Fields is utilizing Solameter 9.4 to directly measure in vitro sunscreen product protection against the blue light portion of solar light. The relevant data are being collected and analyzed to determine the feasibility of this approach. Thus far, 21 commercial sunscreen products were evaluated using this technique and it was found that their blue light/UV ratios differ.
Dueva-Koganov explained that mineral sunscreen actives used in conjunction with certain organic sunscreen actives and particulate materials (silica, hydrated silica and iron oxides), contribute to sunscreen’s improved absorbance in the blue light region.
“Sunscreen actives currently under the TEA process at the FDA could help to develop more efficacious sunscreen products with improved performance against UVB/UVA and adjacent blue light,” Dueva-Koganov concluded. “The approval of sunscreen actives under the TEA process would benefit US consumers.”
We’ll have more coverage of the Center for Dermal Research in the coming weeks.
The theme for this year’s conference was “Future of Dermatologicals and Cosmeceuticals.” The meeting was attended by more than 180 participants and over 20 sponsors and donors. This was the highest attendance of any of the previous meetings, according to Michniak-Kohn. Companies represented included Rodan + Fields, Johnson & Johnson, MedPharm, Logan Instruments Corp., Tergus Pharma, Dow Development, Gattefosse, Silab, BASF, Jeen International, Galderma-Nestle Skin Health, GenoSkin, Evonik, Genemarkers, Beri Esthetique, Colgate-Palmolive, L’Oréal, Lubrizol, Novan, Avon Products, PCA Skin and Tioga Research.
There were 21 speakers and a regulatory panel which included a speaker from the FDA, Dr. Nakissa Sadrieh, director of the agency’s cosmetics division. In addition, the poster session attracted more than 20 posters in the skin research areas. Attendees came from all over the US as well as internationally (primarily Europe and South America), according to Michniak-Kohn. The annual award of CDR Sponsor of the Year was awarded to Tergus Pharma.
Startling Statistics
Leading off was a keynote by Dr. David Margolis of the University of Pennsylvania, who noted that in 2013, 85 million Americans, 25% of the population, saw a dermatologist and were treated for 1.6 skin diseases. That same year, $75 billion was spent on skin care treatment; $46 billion was attributable to medical costs (office visits, procedure and test), $15 billion to prescription drugs and $4 billion to vaccines (58%), other skin procedures (42%) and skin cancer screening (0.1%), which corresponds to 3.8% of 2013 total US health care costs. The cost per capita was $240; for comparison, diabetes is $950; cardiovascular disease is $950 and renal failure is $147.
Cost corresponding to OTC products for skin disease was nearly $10 billion. “For OTC, that works out $32 per year per person for skin diseases,” noted Margolis. “Most of the drugs used by dermatologists were not originally implicated for skin diseases.”
On a global scale, skin disease is a leading cause of disease burden. Margolis pointed to a 2014 study in the Journal of Investigative Dermatology which found that three skin conditions—fungal skin diseases, other skin and subcutaneous diseases, and acne—were in the top 10 most prevalent diseases worldwide in 2010, and eight fell into the top 50; these additional five skin problems were pruritus, eczema, impetigo, scabies and molluscum contagiosum. Yet, very few studies, according to Margolis, have directly focused on skin disease.
He reminded the audience that the US and global populations are getting older, which means a growing demand for products to treat chronic wounds such as diabetic foot ulcers, which is the No. 1 cause of amputations in the US. In 1980, there were 5.5 million cases of diabetes; by 2014, the number of cases had climbed to 24 million, according to Margolis, who also noted that people who have amputations are 20% more likely to die in any given year.
Psoriasis and atopic dermatitis also demand attention from doctors and formulators. Margolis noted that psoriasis affects 2-4% of the adult population and is associated with an aging population. In contrast, atopic dermatitis is prevalent in 10-20% of children in industrialized nations and is associated with other atopic illnesses such as seasonal allergies, asthma, and drug and food allergies. No wonder why so many companies are focused on psoriasis and atopic dermatitis.
Novan president Nathan Stasko explained how a 100% nitric oxide treatment developed by his company effectively treats inflammation, acne, atopic dermatitis and psoriasis. The company’s Nitricil technology enables the delivery of nitric oxide by storing the gaseous species on large polymers, which has led to the development of a diverse pipeline of time-release, nitric oxide-releasing new chemical entities for short- or long-term healthcare applications. The result, according to Novan, is a stabilized, drug-able nitric oxide that can be formulated into gels, ointments and creams.
There are a range of dermal penetration enhancers that may be incorporated into topical pharmaceutical formulations, noted Inayet Ellis of Gattefossé; these include hydroalcoholic solvents (alcohols, ether alcohols, glycols); lipophilic solvents (esters, fatty esters) and surfactants (nonionic, anionic and cationic).
“Hydration of the stratum corneum promotes absorption of both hydrophilic and lipophilic materials. Proper use of excipients and formulation design facilitate passage across the stratum corneum and drive diffusion past the stratum corneum,” concluded Ellis. “Safe delivery of medicines via the skin is possible, but the formulator must strike the right balance between solubilization, partitioning and localization.”
A Rise in Pollutants
According to the World Health Organization (WHO), more than 90% of the urban population live with pollutant levels above the WHO-defined standard limits. Yong Zhuang of Avon Products reviewed some of the most common pollutants including polycyclic aromatic hydrocarbons (PAHs), which he called among the most widespread and dangerous organic pollutants. PAHs are converted into quinines, redox-cycling chemicals that produce reactive oxygen species (ROS), which in turn, damage DNA and lead to skin aging. Some of the environmental aggressors that produce oxidative damage to skin include hydrogen peroxide, UVA/B and urban dust. However, Avon researchers identified a blend of antioxidants that can help prevent and/or repair damage caused by a number of environmental aggressors in vitro. Finally, a topical formula (Anew Skinvincible) incorporating this blend of antioxidants has been shown to improve skin health in a clinical study.
Yarka Kepic, Verdi Enterprises Inc., reviewed how efficacy levels of bioactive neuropeptides are impacting neurocosmetic formulations. She noted that these materials are effective at 5 parts per million compared to 700 parts per million for retinol. Other neurocosmetic actives include cinnamaldehyde, monks pepper, pumpkin seed extract and dihydroavenanthramide D. In the not-to-distant-future, these and other actives will improve the efficacy of a range of cosmetic and OTC products including premature aging, sunless tanning, chronic pain and itch, wound healing, acne rosacea and psoriasis, she explained.
“Future dermaceutical and cosmeceutical products will rely on increased use of synthetic biometric peptides,” she concluded. “There will be increased use of sustainable materials which elicit a neurogenic response.”
Retinoids increase fibroblast growth and dermal collagen synthesis, while decreasing levels of MMPs, according to PCA Skin’s Ivana Veljkovic who reviewed the material’s history and mechanism of action in skin formulations.
“Retinoids have been implicated in nearly all phases of epidermal differentiation and keratinization,” she added.
The Role of Retinoids
Veljkovic admitted that initially, acne therapy with retinoids can be frustrating, as in the first few weeks of use, the follicular epithelium loosens and, when mixed with pro-inflammatory lipids in sebum, can lead to increased breakouts. To reduce this negative effect, retinoid are often used in tandem with oral and topical antibacterials, such as benzoyl peroxide. Despite this drawback, retinoids remain the most effective comedolytic agents making them appropriate for comedonal and inflammatory acne treatments, according to Veljkovic. Now, retinoids are thought to have an effect on intrinsic aging.
“This is important, as previously, it was believed that we could only impact extrinsic aging and all intrinsic aging was inevitable,” she explained.
With regular use of retinoids, smoother skin usually occurs in the first four weeks and is likely the result of an increase in epidermal proliferation and consequential epidermal thickening, compaction of the stratum corneum and deposition of glycosaminoglycans in the stratum corneum and in the intercellular spaces of the epidermis. Retinoids reduce pigment levels in skin by inhibiting melanogensis, inhibiting the transfer of melanin from melanocytes to keratinocytes, and increasing the shedding of melanin-laden keratinocytes as a result of epidermal proliferation, explained Veljkovic.
“Despite the benefits of topical retinoids, instability makes it extremely hard to use them effectively in formulations,” she noted.
At the same time, retinoids can be very irritating. To overcome some of these limitations, PCA relies on non-irritating molecules (OmniSomes) to keep retinol protected and stable and increase skin penetration. Another PCA strategy is use of the pro-drug concept, whereby retinol is well-delivered and converted into the biologically active form, retinoic acid, with reduced irritancy. Of course, each formula includes ancillary ingredients to enhance each product’s overall efficacy, she concluded.
PCA has also maximized L-ascorbic acid-based skin care with the launch of C&E Advanced with Hexylresorcinol and Silymarin. According to PCA the formula provides “unparalleled” antioxidant protection by combining four antioxidants that minimize the appearance of lines and wrinkles, reduces hyperpigmentation, protects skin from environmental stressors and strengthens and smooths skin. The formula contains 20% L-ascorbic acid, 5% tocopherol, 1% silymarin and 1% hexylresorcinol.
“(Our) anhydrous delivery technology allows for maximum stabilization of L-ascorbic acid over extended periods of time,” said Veljkovic. “The unique transdermal delivery system allows for full delivery of water-soluble, L-ascorbic acid into the skin over two hours.”
Feeling Blue
Olga Dueva-Koganov, Rodan + Fields, reviewed the latest trends in solar irradiation protection, noting that consumers are becoming increasingly aware of the negative impact of irradiation on the skin beyond UV; e.g., blue light (HEV). In one survey, 28% of Brazilians said they would like to try sun care products that offer protection from visible light. However, existing methods do not explicitly assess the HEV light region.
“To enable the evaluation of sunscreen’s protection potential in the blue light region, we modified Diffy’s equation and utilized it for the evaluation of commercial sunscreen products,” explained Dueva-Koganov. (The modified equation can be found in the slide show in this article.)
Rodan + Fields is utilizing Solameter 9.4 to directly measure in vitro sunscreen product protection against the blue light portion of solar light. The relevant data are being collected and analyzed to determine the feasibility of this approach. Thus far, 21 commercial sunscreen products were evaluated using this technique and it was found that their blue light/UV ratios differ.
Dueva-Koganov explained that mineral sunscreen actives used in conjunction with certain organic sunscreen actives and particulate materials (silica, hydrated silica and iron oxides), contribute to sunscreen’s improved absorbance in the blue light region.
“Sunscreen actives currently under the TEA process at the FDA could help to develop more efficacious sunscreen products with improved performance against UVB/UVA and adjacent blue light,” Dueva-Koganov concluded. “The approval of sunscreen actives under the TEA process would benefit US consumers.”
We’ll have more coverage of the Center for Dermal Research in the coming weeks.