Tom Branna, Editorial Director09.01.21
Nonionic ethoxylates used in skin care products, and the ubiquitous ether sulfates present in so many rinse-off products, are under pressure by regulators and non-government organizations. Claims regarding 1,4-Dioxane emissions have home care and cosmetic manufacturers scrambling to reformulate their fabric and personal care formulas. But industry expert Ricardo Diez would rather fight than switch. In a lively webinar presented by the New York Chapter of the Society of Cosmetic Chemists, Diez outlined the lengths he’s gone to defend this misunderstood class of chemicals.
Diez is an adjunct professor at Rutgers University where he teaches courses within RU’s Masters in Business and Science program. Prior to that, he spent 40 years in the cosmetics industry, working on both the supply and finished product side of the business. Diez noted that rinse-off sulfates, sulfonates and other ethoxylates rose to prominence in personal care formulas due to the presence of oligomer blends.
“The blend of oligomers makes them unique,” he noted. “But bad information, misinformation is appearing all around us.
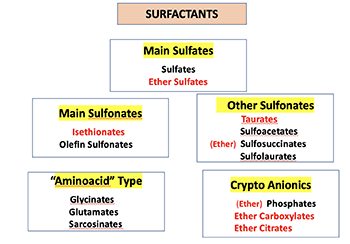
Know your chemistry! Here's an easy way to group surfactants.
“We have one real issue with 1,4 D, and many invented ones. The real problem is that the 1,4 D in rinse-off products from the ether sulfates ends up in the water we drink. An aggravating factor is that, in the US, there are areas where the content is above the limits established by the EPA.
“However, we do not know, or at least I do not know, how much of that 1,4 D comes from cosmetic products, from other consumer products, and from the industrial application of this chemical,” he said. “The invented problems come from only a few sources but they are repeated time and again, as if the sources were many.”
As an example, Diez asked attendees to read the following statement from one NGO that is filled with misinformation regarding shower gels and sodium lauryl ether sulfate.
“During exposure to 1,4 Dioxane from a bath product, a person’s skin is warmed, pores are opened, the skin is soaked in the contaminated water and 1,4 dioxane enters the bloodstream. 1,4 dioxane is also released as a gas and is inhaled as it is trapped in the enclosed area of the bathroom or shower.”
“Do you think that is true?” he asked. “This may surprise the young, but the modern cosmetic industry was based on chemistry! You must prove what you say! Science, chemistry, is not a matter of opinion! The first amendment does not apply! You are not entitled to your opinion. We only care about what you can prove. You prove things through experimentation.”
Diez noted that when he began his career, back in the 1970s, cosmetic chemistry was all about hypotheses and experimentation. But at some point, things began to change—and not for the better.
“In 1995, we got the term ‘Natural.’ In 2005, we got the term ‘Sulfate-free’ and in 2015, we got the term ‘Clean Beauty,’” he recalled.
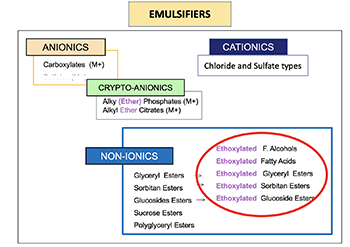
The same idea works for emulsifers, too.
“When I started in the business, the cosmetics industry was part of the chemical industry. You had to prove what you claimed. Today, it’s an industry that makes cosmetics, but it is not part of the chemical industry.”
The Problem with 1,4-Dioxane
Diez insisted that misinformation is at the heart of the issue surrounding 1,4-Dioxane in household and personal products. He explained that in 1985, a spill at a production facility owned by Gelman Science (now Pall Corporation), led to the discovery of 1,4-Dioxane in drinking water wells near the plant. When regulators warned that Gelman would be fined, the company’s defense was “Cosmetic and household products also put 1,4 dioxane in the water supply.”
According to Diez, specific companies (Johnson & Johnson and Procter & Gamble) and brands (Johnson’s Baby Shampoo and Tide) were mentioned by Gelman in its defense.
“I was there,” he recalled. that was the first time that cosmetics, 1,4 dioxane and ether sulfates were linked.”
Despite that link, the industry did nothing. In 2001, the US Food and Drug Administration published a paper that showed 1,4-Dioxane levels rising from 229ppm in 1979 to 348ppm in 1997. Still, industry did nothing.
“If you don’t move your car from a ‘no parking’ zone, sooner or later the cops are going to take it!” observed Diez. “In the 2000s, the presence of 1,4D in the water supplies became more publicized. But the industry did little to control the conversation”
The lack of response by industry to reduce 1,4D levels provided an opening to alternative organizations to “control the conversation.”
“Guess who was driving the tow truck! The Environmental Working Group (EWG) and Safe Cosmetics.org!”
In 2007, the EWG published a study with the headine “Cancer-causing Chemical Found in Children’s Bath Products.” The report also noted that women’s shampoos and body washes also contained 1,4-Dioxane.
EWG went on to say, “To avoid 1,4D, avoid cosmetics with sodium laureth sulfate and ingredients that include ‘PEG,’ ‘xynol,’ ‘ceteareth,’ and ‘oleth.’”
Garbage In, Garbage Out
Like many scientists before him, Diez noted that EWG’s study did not take into account Paracelsus’ observation that “the dose makes the poison.”
In fact, EWG didn’t like the way 1,4-Dioxane looked when measured in parts per million, so it took to reporting 1,4-Dioxane levels in parts per billion for dramatic effect. As a result, Johnson’s Baby Shampoo’s 1,4-Dioxane levels went from 0.87ppm to 870ppb. Similarly, Garnier Fructis with Active Fruit Protein went from 0.4ppm to 400ppb. Among skin care products, Olay Moisture Ribbons Plus Body Wash’s 1,4-Dioxane levels went from 3.5ppm to 3,500ppb and Dove Nutritive Solutions went from 2.2ppm to 2,20ppb.
“If you are a chemist, you know that you cannot just completely invent digits! That is unscientific!” noted Diez. “For dramatic effect, the Campaign for Safe Cosmetics called cosmetics an industry of death all based on the presence of dioxane in common ingredients. It is misinformation, misquotations and speculation!”
The Campaign for Safe Cosmetics went on to falsely claim that the levels of 1,4-Dioxane found in many personal care products are 1,000 times higher than those found to cause cancer in laboratory animals. In another misinformation campaign, the Campaign for Safe Cosmetics falsely claimed: “For example, during exposure to 1,4-Dioxane from a bath product, a person’s skin is warmed, pores are opened, the skin is soaked in the contaminated water, and 1,4-Dioxane enters the bloodstream. 1,4-Dioxane is also released as a gas and is inhaled as it is trapped in the enclosed area of the bathroom or shower.”
“If it were true, we would all be dead!” noted Diez.
Fighting Back
Diez recalled decades of safety research conducted by Unilever, Procter & Gamble and other household and personal products companies. But he noted that upstart companies, armed with misinformation from EWG and others, could gain market share by marketing sulfate-free personal care formulas. He told webinar attendees that the sulfate-free movement could have died by itself or remained isolated to a limited, high-end salon market.
“Unfortunately, some sectors of the industry realized that sulfate-free products could be a way to enter or compete on the crowded shampoo shelves,” he recalled. “The veracity of the claims against the sulfate products was irrelevant. Now, newcomers could compete against P&G and Unilever, whether their claims were true or not.”
But Diez isn’t backing down from his defense of ethoxylated ingredients.
Last month, he emailed a manufacturer of sulfate-free and asked: “Could you please clarify for me why coco sulfate is good and lauryl sulfate is not?”
The response came back: “Sodium Coco Sulfate is verified by EWG as safe and non-hazardous to health.”
Another email followed: “Please do not confuse sodium lauryl sulfate and sodium coco sulfate with sodium laureth sulfate (SLES). SLES is synthesized from SLS via ethoxylation and this process can generate 1,4-dioxane, a possible human carcinogen, according to the International Agency for Research on Cancer and a carcinogenic substance according to the OEHHA. For this reason, we will never use SLES in any of our products.”
Next, Diez took on the EWG asking for details on the process it uses to assess product safety. EWG responded by guiding Diez to the group’s Skin Deep page.
Unfortunately, the page only gives a numerical ranking for individual molecules without knowing the physical structures that are formed when they come into contact with other ingredients in the formula. Furthermore, EWG’s ranking doesn’t seem to have a handle on basic chemistry.
On July 7, Diez emailed EWG again:
“Reviewing your information on several surfactants, I noticed that in the case of Laureth Sulfates you mention that they can be contaminated with Ethylene Oxide and 1,4 Dioxane. However, you do not make the same comments for Isethionate and Taurate surfactants. Both are also made using Ethylene Oxide.”
How does one ingredient get good marks from EWG and another get poor marks? As The EcoWell pointed out, NGOs are heavily funded by the organic lobby.
“Certifications are not cheap but even larger companies are jumping on the bandwagon,” according to The EcoWell.
Or as Diez noted, “How does it work? If you got a 4, put some money in the machine and you get a 1.”
He pointed out that the American Council on Science and Health blasted the EWG in a column as far back as 2017 (https://www.acsh.org/news/2017/05/25/dear-ewg-why-real-scientists-think-poorly-you-11323).
For example, in 2015, EWG claimed one million pounds of 1,4-Dioxane were produced/imported into the US as per EPA and 675,000 pounds were released into the environment. However, what the EPA actually reported was the current production is approximately 1 million pounds. 61,907 pounds were released into surface water, 486,124 pounds were transferred to off-site disposal. 55,732 pounds were released into the air and 13,370 to onsite land disposal.
“Why are you lying? Why are you lying?” Diez asked.
He noted that 1,4-Dioxane is formed by a chemical reaction that requires an acid medium to happen. The ethoxylation step is done with an alkaline catalyst, hence ethoxylated emulsifiers have very little 1,4-Dioxane which can be distilled-off under vacuum. On the other hand, ether sulfates have more 1,4 D because the sulfonation process is done in a highly acidic medium, In addition, you need specialized equipment to remove it.
Diez noted that EWG complained that the FDA disregarded one paper that shows that 1,4D is adsorbed by the skin.
“Don’t give me that crap! I work in a University and I can find any paper,” jeered Diez.
The paper in question was published in Toxicology in Vitro. Vol. 27, Issue 2. March 2013. Pages 708-713. “
In that paper, titled "Studies in percutaneous penetration of chemicals—impact of storage conditions for excised human skin, researchers applied an “infinite doses of of neat 1,4D were applied onto the upper side of the skin and the exposure chamber was occluded.”
To which Diez, replied, “Of course, if you have an infinite dose and it is occluded so it has nowhere to go, of course it is going into the skin!”
In conclusion, Diez noted that cosmetics industry started as part of the chemical industry.
“I don’t know what requirements you need to start a cosmetics company today,” he concluded. “Do you just follow the recipe given to you by a supplier and then claim whatever you want?”
Diez is an adjunct professor at Rutgers University where he teaches courses within RU’s Masters in Business and Science program. Prior to that, he spent 40 years in the cosmetics industry, working on both the supply and finished product side of the business. Diez noted that rinse-off sulfates, sulfonates and other ethoxylates rose to prominence in personal care formulas due to the presence of oligomer blends.
“The blend of oligomers makes them unique,” he noted. “But bad information, misinformation is appearing all around us.

Know your chemistry! Here's an easy way to group surfactants.
“However, we do not know, or at least I do not know, how much of that 1,4 D comes from cosmetic products, from other consumer products, and from the industrial application of this chemical,” he said. “The invented problems come from only a few sources but they are repeated time and again, as if the sources were many.”
As an example, Diez asked attendees to read the following statement from one NGO that is filled with misinformation regarding shower gels and sodium lauryl ether sulfate.
“During exposure to 1,4 Dioxane from a bath product, a person’s skin is warmed, pores are opened, the skin is soaked in the contaminated water and 1,4 dioxane enters the bloodstream. 1,4 dioxane is also released as a gas and is inhaled as it is trapped in the enclosed area of the bathroom or shower.”
“Do you think that is true?” he asked. “This may surprise the young, but the modern cosmetic industry was based on chemistry! You must prove what you say! Science, chemistry, is not a matter of opinion! The first amendment does not apply! You are not entitled to your opinion. We only care about what you can prove. You prove things through experimentation.”
Diez noted that when he began his career, back in the 1970s, cosmetic chemistry was all about hypotheses and experimentation. But at some point, things began to change—and not for the better.
“In 1995, we got the term ‘Natural.’ In 2005, we got the term ‘Sulfate-free’ and in 2015, we got the term ‘Clean Beauty,’” he recalled.

The same idea works for emulsifers, too.
The Problem with 1,4-Dioxane
Diez insisted that misinformation is at the heart of the issue surrounding 1,4-Dioxane in household and personal products. He explained that in 1985, a spill at a production facility owned by Gelman Science (now Pall Corporation), led to the discovery of 1,4-Dioxane in drinking water wells near the plant. When regulators warned that Gelman would be fined, the company’s defense was “Cosmetic and household products also put 1,4 dioxane in the water supply.”
According to Diez, specific companies (Johnson & Johnson and Procter & Gamble) and brands (Johnson’s Baby Shampoo and Tide) were mentioned by Gelman in its defense.
“I was there,” he recalled. that was the first time that cosmetics, 1,4 dioxane and ether sulfates were linked.”
Despite that link, the industry did nothing. In 2001, the US Food and Drug Administration published a paper that showed 1,4-Dioxane levels rising from 229ppm in 1979 to 348ppm in 1997. Still, industry did nothing.
“If you don’t move your car from a ‘no parking’ zone, sooner or later the cops are going to take it!” observed Diez. “In the 2000s, the presence of 1,4D in the water supplies became more publicized. But the industry did little to control the conversation”
The lack of response by industry to reduce 1,4D levels provided an opening to alternative organizations to “control the conversation.”
“Guess who was driving the tow truck! The Environmental Working Group (EWG) and Safe Cosmetics.org!”
In 2007, the EWG published a study with the headine “Cancer-causing Chemical Found in Children’s Bath Products.” The report also noted that women’s shampoos and body washes also contained 1,4-Dioxane.
EWG went on to say, “To avoid 1,4D, avoid cosmetics with sodium laureth sulfate and ingredients that include ‘PEG,’ ‘xynol,’ ‘ceteareth,’ and ‘oleth.’”
Garbage In, Garbage Out
Like many scientists before him, Diez noted that EWG’s study did not take into account Paracelsus’ observation that “the dose makes the poison.”
In fact, EWG didn’t like the way 1,4-Dioxane looked when measured in parts per million, so it took to reporting 1,4-Dioxane levels in parts per billion for dramatic effect. As a result, Johnson’s Baby Shampoo’s 1,4-Dioxane levels went from 0.87ppm to 870ppb. Similarly, Garnier Fructis with Active Fruit Protein went from 0.4ppm to 400ppb. Among skin care products, Olay Moisture Ribbons Plus Body Wash’s 1,4-Dioxane levels went from 3.5ppm to 3,500ppb and Dove Nutritive Solutions went from 2.2ppm to 2,20ppb.
“If you are a chemist, you know that you cannot just completely invent digits! That is unscientific!” noted Diez. “For dramatic effect, the Campaign for Safe Cosmetics called cosmetics an industry of death all based on the presence of dioxane in common ingredients. It is misinformation, misquotations and speculation!”
The Campaign for Safe Cosmetics went on to falsely claim that the levels of 1,4-Dioxane found in many personal care products are 1,000 times higher than those found to cause cancer in laboratory animals. In another misinformation campaign, the Campaign for Safe Cosmetics falsely claimed: “For example, during exposure to 1,4-Dioxane from a bath product, a person’s skin is warmed, pores are opened, the skin is soaked in the contaminated water, and 1,4-Dioxane enters the bloodstream. 1,4-Dioxane is also released as a gas and is inhaled as it is trapped in the enclosed area of the bathroom or shower.”
“If it were true, we would all be dead!” noted Diez.
Fighting Back
Diez recalled decades of safety research conducted by Unilever, Procter & Gamble and other household and personal products companies. But he noted that upstart companies, armed with misinformation from EWG and others, could gain market share by marketing sulfate-free personal care formulas. He told webinar attendees that the sulfate-free movement could have died by itself or remained isolated to a limited, high-end salon market.
“Unfortunately, some sectors of the industry realized that sulfate-free products could be a way to enter or compete on the crowded shampoo shelves,” he recalled. “The veracity of the claims against the sulfate products was irrelevant. Now, newcomers could compete against P&G and Unilever, whether their claims were true or not.”
But Diez isn’t backing down from his defense of ethoxylated ingredients.
Last month, he emailed a manufacturer of sulfate-free and asked: “Could you please clarify for me why coco sulfate is good and lauryl sulfate is not?”
The response came back: “Sodium Coco Sulfate is verified by EWG as safe and non-hazardous to health.”
Another email followed: “Please do not confuse sodium lauryl sulfate and sodium coco sulfate with sodium laureth sulfate (SLES). SLES is synthesized from SLS via ethoxylation and this process can generate 1,4-dioxane, a possible human carcinogen, according to the International Agency for Research on Cancer and a carcinogenic substance according to the OEHHA. For this reason, we will never use SLES in any of our products.”
Next, Diez took on the EWG asking for details on the process it uses to assess product safety. EWG responded by guiding Diez to the group’s Skin Deep page.
Unfortunately, the page only gives a numerical ranking for individual molecules without knowing the physical structures that are formed when they come into contact with other ingredients in the formula. Furthermore, EWG’s ranking doesn’t seem to have a handle on basic chemistry.
On July 7, Diez emailed EWG again:
“Reviewing your information on several surfactants, I noticed that in the case of Laureth Sulfates you mention that they can be contaminated with Ethylene Oxide and 1,4 Dioxane. However, you do not make the same comments for Isethionate and Taurate surfactants. Both are also made using Ethylene Oxide.”
How does one ingredient get good marks from EWG and another get poor marks? As The EcoWell pointed out, NGOs are heavily funded by the organic lobby.
“Certifications are not cheap but even larger companies are jumping on the bandwagon,” according to The EcoWell.
Or as Diez noted, “How does it work? If you got a 4, put some money in the machine and you get a 1.”
He pointed out that the American Council on Science and Health blasted the EWG in a column as far back as 2017 (https://www.acsh.org/news/2017/05/25/dear-ewg-why-real-scientists-think-poorly-you-11323).
For example, in 2015, EWG claimed one million pounds of 1,4-Dioxane were produced/imported into the US as per EPA and 675,000 pounds were released into the environment. However, what the EPA actually reported was the current production is approximately 1 million pounds. 61,907 pounds were released into surface water, 486,124 pounds were transferred to off-site disposal. 55,732 pounds were released into the air and 13,370 to onsite land disposal.
“Why are you lying? Why are you lying?” Diez asked.
He noted that 1,4-Dioxane is formed by a chemical reaction that requires an acid medium to happen. The ethoxylation step is done with an alkaline catalyst, hence ethoxylated emulsifiers have very little 1,4-Dioxane which can be distilled-off under vacuum. On the other hand, ether sulfates have more 1,4 D because the sulfonation process is done in a highly acidic medium, In addition, you need specialized equipment to remove it.
Diez noted that EWG complained that the FDA disregarded one paper that shows that 1,4D is adsorbed by the skin.
“Don’t give me that crap! I work in a University and I can find any paper,” jeered Diez.
The paper in question was published in Toxicology in Vitro. Vol. 27, Issue 2. March 2013. Pages 708-713. “
In that paper, titled "Studies in percutaneous penetration of chemicals—impact of storage conditions for excised human skin, researchers applied an “infinite doses of of neat 1,4D were applied onto the upper side of the skin and the exposure chamber was occluded.”
To which Diez, replied, “Of course, if you have an infinite dose and it is occluded so it has nowhere to go, of course it is going into the skin!”
In conclusion, Diez noted that cosmetics industry started as part of the chemical industry.
“I don’t know what requirements you need to start a cosmetics company today,” he concluded. “Do you just follow the recipe given to you by a supplier and then claim whatever you want?”
















