Tom Branna, Editorial Director10.01.18
Does your product perform as promised? Will your laundry detergent get out grass stains? Will your night cream reduce the appearance of wrinkles? More importantly, especially when it comes to cosmetics, is your product safe and are your ingredients “clean?” These issues have come to the fore in the past year as regulators debate the merits of cosmetic regulation bills and local law enforcement agencies encounter counterfeit products.
In April, the LAPD confiscated $700,000 worth of counterfeit makeup that tested positive for human waste contamination. The article made headlines in news outlets around the world and underscored the need for product testing and, of course, sanitary manufacturing conditions. Obviously, counterfeiters are on the wrong side of the law, but there are plenty of law-abiding startups who may not test their ingredients or finished products and end up with contamination problems or efficacy issues.
And that’s where testing companies flex their muscles.
“The most common requests concern the evaluations of SPF and anti-aging; such as anti-wrinkle, firmness, hydration, prevention of free radicals,” observed Anne Sirvent, scientific communication, R&D manager, Dermscan. “These are claims that are found on almost all products; consumers easily understand them, are familiar with them and look for them on packaging.”
Sensitive Types
According to Comprehensive Research Group CEO Tracey Baubie, demand has increased for specialized skin care product testing; namely, an upturn in testing populations with specific skin attributes, such as eczema, hyperpigmentation, and specific age-related attributes, such as dark circles, crow’s feet or under eye wrinkles.
“More and more, industry leaders are not merely seeking subject populations with ‘dry’ or ‘aging’ skin, but they are pin-pointing an issue of concern for skin care consumers and creating specific products to address those concerns,” explained Baubie. “With this narrowing, comes a heightened demand for a more specialized subject population. Thankfully, we have been successful in meeting these demands.”
She pointed toward significant growth in a couple of different areas. First, on-going consumer interest in “natural,” “organic,” or “clean” beauty and personal care products has exploded as consumers seek products that are free from synthetic fragrances, preservatives, parabens and petrochemicals. According to Baubie, while there are certainly consumers who will use “clean” products without proof of effectiveness, many others will lean clean only when the product is backed by clinical proof of its effectiveness.
“Consequently, we are seeing a heightened call for the rigorous testing of ‘clean’ product lines to clinically demonstrate that such products are just as effective (or more so) than traditionally produced product lines,” said Baubie. “We believe that the Millennials will continue to drive the ‘clean’ beauty and personal care market. As such, we expect to see a huge uptick in growth for these products during the next decade and beyond. We also anticipate R&D expenditures will grow accordingly.”
Furthermore, Baubie noted that DNA/RNA analysis is becoming more common in beauty and personal care product testing. After all, at-home DNA testing kits now purport to help identify the health of a consumer’s skin and hair, predicting how soon, for example, you will gray or how your skin reacts to UVA/UVB rays, she explained.
“Again, we think this is a consumer-driven market that will require substantial clinical testing, as the concept of highly-individualized personal and beauty care products continues to gain more traction in the years to come,” predicted Baubie. “In the not-too-distant future, DNA testing may also have an impact on more targeted recruitment of subjects with specific genetic makeup.”
Blue Light Specials
The elements can wreak havoc on skin. Cosmetic chemists have known the deleterious effects of UVB for decades; more recently, the damage caused by UVA have come to the fore. Now, pollution and blue light claims are de rigueur. Testing companies must remain ahead of the curve when it comes to today’s product claims and promises.
“We regularly offer new protocols to meet the market trends and answer the needs of our customers,” explained Sirvent. “Dermscan has more than 20 years of experience in anti-pollution testing. We propose tests on the prevention of fine particles deposit or the cleansing effect against these, either on the skin or the hair.”
The company can also conduct research on prevention of heavy metals deposition and anti-free radical effects in connection with urban pollution and cigarette smoke.
In the quest for complete solar protection, Dermscan tests in vivo the impact of blue light and infra-red. The microbiota is also an emerging trend for which the company recommends specific protocols for claims on the maintenance of cutaneous flora (for healthy skin) or on the analysis of specific strains in the case of cutaneous disorders (dryness, skin with a tendency to acne, film, deodorants, etc.).
“We also propose to follow the impact of cosmetics on well-being and quality of life thanks to targeted questionnaires, coupled with product effectiveness monitoring,” said Sirvent. “We have created specific protocols for ‘anti-fatigue’ and ‘detoxifing’ claims.”
Finally, Dermscan developed a specific protocol for makeup that responds to the “athleisure” trend; i.e., long-lasting and sweat-resistant makeup.
You Animal!
Animal safety testing of consumer products started getting bad press back in the 1980s. Protesters dressed as “draized” rabbits, received valuable airtime and made headlines in the US and around the world, sparking the no animal testing movement within the cosmetics industry. Back then, in vitro testing of cosmetics and household cleaning products was in its infancy, but InVitro International, Placentia, CA, was on the scene issuing a clarion call for change.
Company CEO Rich Ulmer is still at it, and he is convinced that the tide has turned in favor of in vitro methods. He said the Organisation for Economic Co-operation and Development (OECD) is closer to approving InVitro’s Ocular Irritection test. According to In Vitro International, OECD is generally viewed as the world’s foremost regulatory authority on in vitro test methods.
“We see growing interest in Ocular Irritection in Europe,” explained Ulmer. “We will be there in November, we want to see it across the goal line. It is our No. 1 priority.”
Europe isn’t the only region interested in animal testing alternatives. Ulmer called Brazil a “huge opportunity” for non-animal testing.
“My sources say that Brazil will minimize animal tests by 2020,” he told Happi. “That’s the warning shot that’s been fired.”
Brazil is following in the footsteps of countries such as India and Taiwan, which banned animal testing for cosmetics in 2013 and 2016, respectively. Now, California, is poised to do the same. Last month, California State Assembly passed bill SB 1249, which makes it illegal to sell products that have been, or include ingredients that have been, tested on animals. If signed into law by Governor Jerry Brown, the change will go into effect on Jan. 1, 2020, and would apply to all brands currently selling in California as well as any others that enter the market.
The Independent Cosmetic Manufacturers and Distributors (ICMAD) noted inconsistencies by California legislators, as the state advocates animal testing by accepting data from animal tests to determine if a chemical/ingredient should be listed on its Proposition 65 as a possible carcinogen or reproductive toxicant. So, while with this legislation, California is looking to ban animal testing, with Prop 65, it is requiring animal testing.
“Make no mistake, no one is advocating that animal testing be continued. What we do believe is that attacking our industry is the wrong approach,” concluded ICMAD. “We believe this issue is one that needs to be addressed directly by supporting the development of alternate testing models and reference data bases and not by passing laws that are purely sensational and have no real effect. ICMAD will keep working towards national uniformity on this issue and others.”
For its part, ICMAD supports the Safe Cosmetics Modernization Act (HR575), a bill introduced by Rep. Pete Sessions (R-TX). The bill would require registration of cosmetic manufacturers, reporting on cosmetic ingredients and reporting of adverse events, but includes no provisions for FDA regulation.
ICMAD isn’t alone in calling for national uniformity on cosmetic regulation. Two bills are stuck in the US Senate that would revamp the US Food and Drug Administration’s ability to regulate cosmetics. In May, 2017, Senators Dianne Feinstein (D-CA) and Susan Collins (R-ME), reintroduced the Personal Care Products Safety Act (S1113), which would require the FDA to review five cosmetic ingredients a year and would specify the first five. A year ago, Senator Orrin Hatch (R-UT) introduced S2003, a bill that NGOs call a “less ambitious alternative” to S1113.
According to Baubie, while both Senate bills would require mandatory registration of cosmetic facilities, ingredient reporting and review, only the Feinstein-Collins bill would give the FDA authority to order product recalls and the authority to annually investigate the safety of specified ingredients. The Feinstein-Collins bill also includes a “fee” provision aimed at providing the resources needed to fund the FDA’s proposed oversight.
“Any new regulations will no doubt have an industry impact,” noted Baubie. “It is not clear, however, what that impact will be, and it will remain unclear until the legislation is refined into law. While one might expect that such legislation would be opposed by the major industry players, that does not appear to be case. Several of the largest beauty and personal care product brands have confirmed their public support for such legislation.”
But whatever proposal you prefer, Arent Fox attorneys don’t expect any movement on either bill prior to the seating of the next Congress.
Importantly, neither bill was able to move forward towards passage and demonstrate significant support in Congress at this time. Thus, the next Congress will be the next opportunity to introduce such legislation and work for passage, according to Georgia C. Ravitz, partner, Arent Fox, a Washington, DC law firm which specializes in the cosmetic industry.
“We recognize that there are a number of committed members in Congress like Feinstein who are committed to increasing industry oversight and will continue to look for opportunities to move such legislation forward,” said Ravitz. “But given the anti-regulation climate in DC, we don’t believe the current Congress and Administration are sufficiently supportive of passing new regulations of this type unless a major safety incident were to occur to change the landscape for cosmetics reform. However, a new Congress will begin in January 2019 and we will look closely then to assess the possibilities and priorities that materialize.”
When Congress finally takes up the issue of cosmetic reform, it should provide a lift for testing companies—and new costs for cosmetic companies and their suppliers, as industry experts predict any new regulations will include, an annual user fee similar to what is currently in place at FDA for drugs and devices.
“Ostensibly, the user fees would be used by FDA to increase staff so the agency could expand its safety compliance programs and related enforcement and regulatory oversight,” said Naomi Halpern, counsel, Arent Fox.
Given the new regulatory requirements included in each bill, the impacts on industry would include increased ingredient and finished product testing to substantiate ingredient/product safety, according to Halpern. Cosmetic GMPs would also increase facility compliance costs, particularly for smaller facilities that don’t have the resources to devote to expanding GMP compliance, such as equipment calibration, retrofitting, increased record keeping, reporting and safety substantiation testing, she added.
Arent Fox experts note that the FDA Cosmetic Safety and Modernization Act and the Personal Care Products Safety Act are largely similar to the comprehensive cosmetic reform proposals that have been introduced in each recent Congress going back more than 10 years—but neither proposal has ever really had enough support for passage. Similar to past measures, these bills include provisions that would require: facility registration; submission of cosmetic ingredient statements; adverse event reporting; the establishment of cosmetic GMPs; record keeping; and greater focus on ingredient safety and the role of non-functional ingredients, among other things, according to James Hartten, senior public affairs and regulatory analyst, Arent Fox.
“Both bills were introduced in 2017 and referred to the Senate Health Committee. We see the prospects for passage of a broad cosmetics reform measure as very low in the current Congress, as there is very little time left in this session, and with this being an election year and the current anti-regulation climate in DC. Plus, since introduction, neither bill has been moved forward in Committee,” explained Hartten. “So really no chance of passing this session and both measures will sunset at the end of 2018. However, similar legislation will likely be introduced again in 2019 and if either the Senate of House were to flip the chances of passage of a cosmetics reform measure would certainly improve.”
According to Arent Fox, these bills increase the focus on cosmetic ingredient safety and being able to substantiate the safety of both ingredients and finished products, including having testing data to support product/ingredient safety. Were such a bill to be enacted, it is unlikely that testing companies would be adversely affected and may see greater demand for testing services.
And while the Feinstein-Collins bill would give the FDA authority to order product recalls and the authority to annually investigate the safety of specified ingredients, Arent Fox attorneys pointed out that based on their experience, companies are often willing to conduct voluntary recalls when legitimate safety issues exists, so the practical consequence of greater FDA recall authority may be not be impactful. This bill also directs FDA to identify a small number of ingredients each year that would be investigated for safety—a little like a Cosmetic Ingredient Review (CIR), which could be utilized in an advisory role should this type of review process ever to be implemented, according to Arent Fox.
Global Aspirations
While the world waits on Washington, testing companies continue to expand their testing services into new product categories and geographic regions. Dermscan offers tests on all types of population, climates and cosmetic habits. For these reasons, the company is already present in Europe (France and Poland) as well as in Thailand and Tunisia. To complete its offer, last year, Dermscan established a working relationship with Insight Research, Mauritius.
Wherever testing takes place, Sirvent pointed out that each region of the world has its own regulations and CROs should have an expert knowledge of each one in order to advise its customers about their best options.
“Thus, in the US, the conditions for evaluating the SPF are not the same as in Europe,” she said. “In France, the law ‘Jardé’ supervises clinical tests and it is important to take it into account, even in the context of a study on a cosmetic product.”
CRG is continuously building its capabilities to keep pace with the growth demands of its industry sponsors, according to Baubie. For example, the company met industry demands by building a state-of-the-art hot room for antiperspirant studies, training and certifying malodor assessors. It regularly engages specialized medical personnel as the need for dermatologists, ophthalmologists, neo-natal and pediatric professionals arise. This year is no exception. Industry innovation in hair care products is in high-demand, so this year CRG has focused on expanding its hair expertise.
“We have recently broadened our hair assessment capabilities to include certified hair graders in hair color, tone, texture, condition, flexibility, shine, and percentage gray,” explained Baubie.
“Additionally, we are expanding our expertise in dandruff grading. CRG has one of the most experienced and sought-after dandruff graders in the country and we want to continue to build on that expertise by ensuring that we have a second generation of dandruff graders who have been rigorously trained and qualified by a leading expert.”
Finally, while CRG is a Minnesota company, Baubie and her team pride themselves on being able to service client needs, wherever those needs arise.
“We have the demonstrated capability to take our “show-on-the-road,” she said. “We have successfully partnered with other entities both nationally and internationally, to ensure that our expertise extends well-beyond our Minnesota borders. Our willingness and ability to take our know-how on the road, keeps our costs highly competitive, since our overhead remains centralized, rather than exponentially multiplied across several regional sites.”
No matter where a testing company’s headquarters are located, if its clients do business in the US, any regulatory action is expected to create more demand for testing services.
A list of testing companies begins below.
Testing Service Providers
Below is a list of testing providers serving the household and personal products industry. For more information, contact the testing service provider using the information provided for each firm.
Should your company be listed here? Contact Happi to be included in our next testing services directory. Send your request to tbranna@rodmanmedia.com.
• Alliance Technologies, LLC
Monmouth Junction, NJ
Tel: (877) 962-5993
Fax: (732) 438-8265
Email: info@alliancetechgroup.com
Website: www.alliancetechgroup.com
Testing Specialties: Alliance Technologies is a full service, DEA-licensed, and FDA-registered and audited contract laboratory offering a wide range of chemical analysis and material testing services to a diverse, international client base. Our expertise and years of experience enable us to select the correct analytical approaches and generate the accurate data needed to solve your toughest problem.
• AMA Laboratories, Inc.
New City, NY
Tel: (845) 634-4330
Fax: (845) 634-5565
Email: sales@amalabs.com
Website: www.amalabs.com
Testing Specialties: AMA Laboratories conducts state-of-the-art testing in RIPT/Safety, Infrared Testing (IRPF), SPF Determinations, Claim Substantiation with Biophysical Measurements, In-Vitro Studies and unique Matched Scientific Photography (MSP) using AMA’s PhotoGrammetrix (PhGx) System. Advanced studies can enhance documented final report data with our PolyChrommetrix (PcMx) 3D modeling plus corporate and advertising video capabilities. The AMA system offers a proprietary concept of scientifically acquired, non-invasive “before and after” images which can be electronically measured. This unique system allows for matched high resolution photographs to be directly incorporated into our clients’ advertising and marketing materials for a truly sensational presentation format.
• Ametek Brookfield
Middleboro, MA
Tel: (508) 946-6200
Email: MA-MID.info@ametek.com
Website: www.brookfieldengineering.com
• Beta Analytic Inc.
Miami, FL
Tel: (305) 662-7760
Email: lab@radiocarbon.com
Website: www.radiocarbon.com
Testing Specialties: Bio-based content and natural products testing services; high-quality results in only 5-7 business days (faster service available) under standards that include ASTM D6866, CEN 16137, EN 16640, ISO 16620, ISO 19984. Results can be accessed online 24/7 including QA reports and sample photos.
• BioScreen Testing Services, Inc.
Torrance, CA
Tel: (310) 214-0043
Fax: (310) 370-3642
Email: info@bioscreen.com
Website: www.bioscreen.com
Testing Specialties: BioScreen Testing Services is a full service, GMP, human clinical, microbiology and analytical chemistry testing laboratory. BioScreen’s clinical services division conducts product safety and efficacy claim studies using a complete array of scientific instrumentation. The company also has sophisticated image capture and analysis software to provide the highest level of scientific image claim support. BioScreen’s clinical services include safety tests for irritation and sensitivity (HRIPT) on the skin and complete ophthalmologic safety tests. BioScreen specializes in customized studies to support product claims from moisturization, anti-aging, SPF and acne, to specialized scar, cellulite, esthetic and eyelash studies. Also offered are Asian subject panels and consumer use studies. BioScreen offers a complete array of analytical chemistry to serve OTC/cosmetic clients from raw material ingredients, sunscreen assay and heavy metals to photo stability, stability, and toxicological assessments. Our microbiological department conducts a compliment of specialized micro studies and ID methods plus more routine preservative testing, microbial plate count, log reduction and bacteriostasis studies.
• Bria Research Labs and Testing Services
Libertyville, IL
Tel: (847) 918-1774
Fax: (847) 816-1830
Email: arun@brialab.com
Website: www.brialab.com
• Canfield Scientific, Inc.
Parsippany, NJ
Tel: (973) 434-1200 or (800) 815-4330
Email: info@canfieldsci.com
Website: www.canfieldsci.com
• Ceutical Laboratories, Inc.
Farmers Branch, TX
Tel: (972) 241-8374
Fax: (972) 241-0619
Email: info@ceuticallabs.com
Website: www.ceuticallabs.com
• Charles River
Charleston, SC
Tel: 877.CRIVER.1 (877-274-8371)
Website: www.criver.com
• Clinical Research
Laboratories, LLC
Piscataway, NJ
Tel: (732) 981-1616
Fax: (732) 981-0520
Email: info@crlresearchlabs.com
Website: www.CRLResearchLabs.com
Testing Specialties: Clinical Research Laboratories, LLC (CRL) operates as an independent contract laboratory providing a wide range of clinical safety and efficacy testing to the cosmetic, beauty products, personal care and pharmaceutical industries. Located both in central New Jersey and Winston-Salem, North Carolina, CRL is dedicated to conducting human clinical test procedures to determine the safety and efficacy of cosmetic, personal care, and OTC drug products. The company focuses its clinical expertise in the areas of dermatology, photobiology, ophthalmology, bioinstrumentation, microbiology, cosmetology, clinical trials and associated services.
• Comprehensive Research Group, Inc.
Minneapolis, MN
Tel: (612) 781-3400
Email: tbaubie@crginc.org
Website: crginc.org
Testing Specialties: For over 20 years, CRG has provided quality data to personal and beauty care manufacturers. We provide superior subject recruitment, study execution and data collection for product development, efficacy, and claims. We have extensive expertise in hair care, skin care, anti-aging treatments, antiperspirants/deodorants, baby care, grooming and safety-in-use testing.
• Consumer Product Testing Company, Inc.
Fairfield, NJ
Tel: (973) 609-5747
Fax: (973) 253-4059
Email: sales@cptcplabs.com
Website: www.cptclabs.com
Testing Specialties: CPTC has provided a full service GMP/GLP/GCP testing laboratory for the cosmetic, personal care, medical device, pharmaceutical and specialty chemical markets. Our capabilities include services in the clinical safety and efficacy, chemistry/microbiology/stability, photobiology, in-vitro toxicological fields and consultation. Our focus is on providing “Conception to Completion” service. We are proud to announce the addition of “Gluten Free” testing, FDA Import Detention testing and new “Bundle” pricing on Predictive Patch, Preservative and Ocular Irritation studies.
• Dermatest Medical Research
Münster, Germany
Tel: (49) 251-481637-0
Fax: (49) 251-481637-66
Email: info@dermatest.de
Website: www.dermatest.de
• Dermscan Group
Lyon, France
Tel: +33(0) 472 823 656
Email: commercial@dermscan.com
Website: www.dermscan.com
Testing Specialties: Dermscan is the reference in clinical trials of cosmetics and pharmaceutical products since 1990 and also realize pre-clinical in vitro tests in GLP certified laboratory. ISO 9001:2015 certified, its facilities in France, Poland, Tunisia, Thailand, Mauritius and international partnerships, provide them multi ethnic panels for standard or complex multiparametric studies.
• EAG Laboratories
San Diego, CA
Tel: (877) 709-9526
Website: www.eag.com
• Essex Testing Clinic, Inc.
Verona, NJ
Tel: 973-857-9541
Email: info@essextesting.com,
Don@essextesting.com
Website: www.essextesting.com
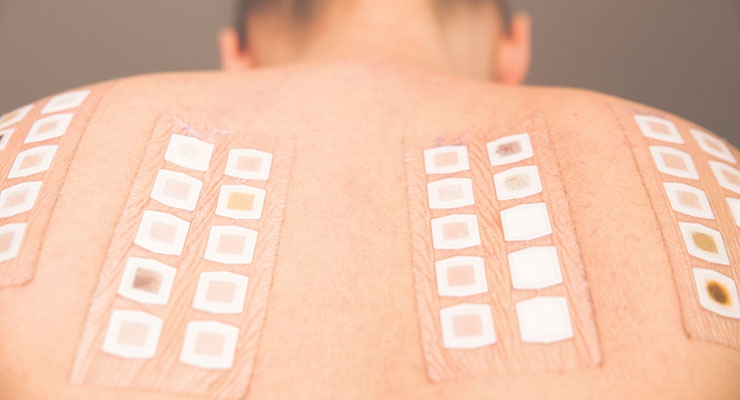
Testing Specialties: Human clinical evaluations performed for safety, efficacy and claims support of cosmetics, raw materials, toiletries, personal care, household, and dermal OTCs. With two facilities and a diverse subject base we specialize in full range of tests, including but not limited to RIPT, SPF, In-Use, Anti-Aging, Patch, photosensitivity, Long Lasting, Waterproof, Comedogenicity, eye products, clinical and perception studies. Protocols developed for specific needs.
• Evalulab Inc.
Montreal, QC, Canada
Tel: (514) 343-0001
Website: www.evalulab.com
Sales: ekulig@evalulab.com
Testing Specialties: Evalulab is a contract research organization offering standard and customized testing services to evaluate safety, performance and consumer preference of skin care, cosmetics, personal care products, medical devices and raw materials. The laboratory is certified ISO 9001:2015 and provides clinical studies that comply to international GCP, FDA and Health Canada.
• Genemarkers, LLC
Kalamazoo, MI
Tel: (844) 220-6231
Email: info@genemarkersllc.com
Website: www.genemarkersllc.com
Testing Specialties: Genemarkers is a contract testing lab that specializes in genomics. Core services include genotyping, gene expression and microRNA profiling. The technology is used to identify the biological mechanisms of action of raw materials and finished products. Testing is used to optimize product development and to validate efficacy.
• Harrison Research
Laboratories, Inc.
Union, NJ
Tel: (908) 688-7600
Fax: (908) 688-7601
Email: client@hrlabs.us.com
Website: www.hrlabs.us.com
• Hill Top Research Incorporated
St. Petersburg, FL
Tel: (727) 344-7602
Email: info@hill-top.com
Website: www.hill-top.com
• Idea Tests Group
Martillac, France
Tel: +33 556 64 82 33
Contact: Frederic Nunzi
Email: f.nunzi@groupeideatests.com
Website: www.groupeideatests.com
Testing Specialties: Idea Tests Group specializes in the evaluation of cosmetic and personal care products for both ingredients and final formulations. Idea Tests Group provides a holistic approach and a wide range of services: in vitro OECD toxicology, efficacy, and microbiology tests, clinical studies to assess tolerance and efficacy, regulatory expertise to support your specific situation, in vitro and in vivo suncare product testing. With seven evaluation centers Idea Tests Group provides its services to customers all over the world.
• Impact Analytical
Midland, MI
Tel: (855) 427-6583
Email: info@impactanalytical.com
Website: www.ImpactAnalytical.com
• International Research Services, Inc.
Port Chester, NY
Tel: (914) 937-9483, (800) 548-0158
Email: info@irsi.org
Website: www.irsi.org
• InVitro International
Placentia, CA 92870
Tel: (800) 246-8487, (949) 851-8356
Fax: (949) 851-4985
Email: invitro@invitrointl.com
Website: www.invitrointl.com
Testing Specialties: Least cost, fastest, validated as accurate—Ocular Irritection and Dermal Irritection. Final reports in as few as 36 hours; accuracy proven as high as 90+% vs animal results. More than a quarter century of satisfied customers with a published peer reviewed OECD study result available. GHS accepted Corrositex: least cost, fastest, and most accurate global leader in non-animal corrosion testing; results possible in 24 hours or less.
• MB Research Laboratories
Spinnerstown, PA
Tel: 215-536-4110
Fax: 215-536-1816
Email: customerservice@mbresearch.com
Website: www.mbresearch.com
Testing Specialties: MB Research Labs has been dedicated to conducting cosmetic and personal care safety assessments for over 40 years. Specialized services include, but are not limited to, in vitro dermal and eye irritation, dermal sensitization, and phototoxicity. An award-winning recognized leader in the development and use of in vitro/alternative toxicology.
• McCrone Group
Westmont, IL
Tel: (630) 887-7100
Website: www.mccroneassociates.com
• Micro Quality Labs Inc. (MQL Inc.)
Burbank, CA
Tel: (818) 845-0070
Fax: (818) 845-0030
Website: www.microqualitylabs.com
Testing Services: Micro Quality Labs is an ISO 17025 certified lab, established in 2003 and offers a comprehensive range of contract testing services to the cosmetic, nutritional, pharmaceutical, OTC and household products industries. Services include Analytical, Stability Testing (ICH guidelines), Heavy Metals, Microbiology, Method Development and Validation, Environmental Testing, Water Testing, Pesticides/USP, 1,4 Dioxane, Amino Acid Testing and Phthalates.
• Princeton Consumer Research
Princeton, NJ
Tel: (609) 455-1112
Fax: (609) 580-1393
Email: janetervooren@princetonconsumer.com
Website: princetonconsumer.com
Testing Specialties: Princeton Consumer Research is a global consumer product testing company specializing in safety, efficacy and acceptability studies. PCR focuses on the cosmetic, skin, beauty, personal care, and baby care industries. Their global headquarters in Princeton, NJ and their site in St. Petersburg, FL have the largest environmentally controlled rooms and hot rooms in the US. PCR also has 2 additional sites in the UK, and a site in Winnipeg, Canada. Plans include expansion to Asia and Brazil in 2018-2019.
• Product Integrity Laboratory LLC
Landing, NJ
Tel: (973) 770-7707
Fax: (973) 770-7711
Email: info@prodinteglab.com
Website: www.prodinteglab.com
Technical Services: Product development and formulation, quality control and quality assurance, regulatory compliance, production troubleshooting.
• Q Laboratories, Inc.
Cincinnati, OH
Tel: (513) 471-1300
Fax: (513) 471-5600
Email: mgoins@qlaboratories.com
Website: www.qlaboratories.com
Testing Specialties: An FDA registered, cGMP/GLP compliant, ISO/IEC 17025 accredited analytical chemistry and microbiology laboratory. Services include: Method development and validation, microbial detection and enumeration, microbial identification, active ingredient analysis, raw material qualification, antimicrobial efficacy analysis and stability studies.
• SGS
Fairfield, NJ
Tel: (973) 575-5252
Fax: (973) 575-7175
Email: uscts.inquiries@sgs.com
Website: www.sgs.com/cosmetics
Testing Specialties: It’s crucial for cosmetics and personal care products to be effective, stable and safe. SGS’ global quality assurance and performance services can help you ensure the quality, safety, efficacy and regulatory compliance of your products. Laboratory and field operations offer tailor-made solutions to meet your chemical, physical, microbiological and claim support needs.
• Skinobs
France
Tel: +33(0)630089098
Email: contact@skinobs.com
Website: www.skinobs.com
Testing Specialties: Analytical tests, toxicology regulatory, tolerance, use tests, ex vivo, in vitro and in vivo efficacy, sensory analysis for skin care, body care, solar, capillary and makeup.
• Solar Light Company, Inc.
Glenside, PA
Tel: (215) 517-8700
Fax: (215) 517-8747
Email: info@solarlight.com
Website: www.SolarLight.com
In April, the LAPD confiscated $700,000 worth of counterfeit makeup that tested positive for human waste contamination. The article made headlines in news outlets around the world and underscored the need for product testing and, of course, sanitary manufacturing conditions. Obviously, counterfeiters are on the wrong side of the law, but there are plenty of law-abiding startups who may not test their ingredients or finished products and end up with contamination problems or efficacy issues.
And that’s where testing companies flex their muscles.
“The most common requests concern the evaluations of SPF and anti-aging; such as anti-wrinkle, firmness, hydration, prevention of free radicals,” observed Anne Sirvent, scientific communication, R&D manager, Dermscan. “These are claims that are found on almost all products; consumers easily understand them, are familiar with them and look for them on packaging.”
Sensitive Types
According to Comprehensive Research Group CEO Tracey Baubie, demand has increased for specialized skin care product testing; namely, an upturn in testing populations with specific skin attributes, such as eczema, hyperpigmentation, and specific age-related attributes, such as dark circles, crow’s feet or under eye wrinkles.
“More and more, industry leaders are not merely seeking subject populations with ‘dry’ or ‘aging’ skin, but they are pin-pointing an issue of concern for skin care consumers and creating specific products to address those concerns,” explained Baubie. “With this narrowing, comes a heightened demand for a more specialized subject population. Thankfully, we have been successful in meeting these demands.”
She pointed toward significant growth in a couple of different areas. First, on-going consumer interest in “natural,” “organic,” or “clean” beauty and personal care products has exploded as consumers seek products that are free from synthetic fragrances, preservatives, parabens and petrochemicals. According to Baubie, while there are certainly consumers who will use “clean” products without proof of effectiveness, many others will lean clean only when the product is backed by clinical proof of its effectiveness.
“Consequently, we are seeing a heightened call for the rigorous testing of ‘clean’ product lines to clinically demonstrate that such products are just as effective (or more so) than traditionally produced product lines,” said Baubie. “We believe that the Millennials will continue to drive the ‘clean’ beauty and personal care market. As such, we expect to see a huge uptick in growth for these products during the next decade and beyond. We also anticipate R&D expenditures will grow accordingly.”
Furthermore, Baubie noted that DNA/RNA analysis is becoming more common in beauty and personal care product testing. After all, at-home DNA testing kits now purport to help identify the health of a consumer’s skin and hair, predicting how soon, for example, you will gray or how your skin reacts to UVA/UVB rays, she explained.
“Again, we think this is a consumer-driven market that will require substantial clinical testing, as the concept of highly-individualized personal and beauty care products continues to gain more traction in the years to come,” predicted Baubie. “In the not-too-distant future, DNA testing may also have an impact on more targeted recruitment of subjects with specific genetic makeup.”
Blue Light Specials
The elements can wreak havoc on skin. Cosmetic chemists have known the deleterious effects of UVB for decades; more recently, the damage caused by UVA have come to the fore. Now, pollution and blue light claims are de rigueur. Testing companies must remain ahead of the curve when it comes to today’s product claims and promises.
“We regularly offer new protocols to meet the market trends and answer the needs of our customers,” explained Sirvent. “Dermscan has more than 20 years of experience in anti-pollution testing. We propose tests on the prevention of fine particles deposit or the cleansing effect against these, either on the skin or the hair.”
The company can also conduct research on prevention of heavy metals deposition and anti-free radical effects in connection with urban pollution and cigarette smoke.
In the quest for complete solar protection, Dermscan tests in vivo the impact of blue light and infra-red. The microbiota is also an emerging trend for which the company recommends specific protocols for claims on the maintenance of cutaneous flora (for healthy skin) or on the analysis of specific strains in the case of cutaneous disorders (dryness, skin with a tendency to acne, film, deodorants, etc.).
“We also propose to follow the impact of cosmetics on well-being and quality of life thanks to targeted questionnaires, coupled with product effectiveness monitoring,” said Sirvent. “We have created specific protocols for ‘anti-fatigue’ and ‘detoxifing’ claims.”
Finally, Dermscan developed a specific protocol for makeup that responds to the “athleisure” trend; i.e., long-lasting and sweat-resistant makeup.
You Animal!
Animal safety testing of consumer products started getting bad press back in the 1980s. Protesters dressed as “draized” rabbits, received valuable airtime and made headlines in the US and around the world, sparking the no animal testing movement within the cosmetics industry. Back then, in vitro testing of cosmetics and household cleaning products was in its infancy, but InVitro International, Placentia, CA, was on the scene issuing a clarion call for change.
Company CEO Rich Ulmer is still at it, and he is convinced that the tide has turned in favor of in vitro methods. He said the Organisation for Economic Co-operation and Development (OECD) is closer to approving InVitro’s Ocular Irritection test. According to In Vitro International, OECD is generally viewed as the world’s foremost regulatory authority on in vitro test methods.
“We see growing interest in Ocular Irritection in Europe,” explained Ulmer. “We will be there in November, we want to see it across the goal line. It is our No. 1 priority.”
Europe isn’t the only region interested in animal testing alternatives. Ulmer called Brazil a “huge opportunity” for non-animal testing.
“My sources say that Brazil will minimize animal tests by 2020,” he told Happi. “That’s the warning shot that’s been fired.”
Brazil is following in the footsteps of countries such as India and Taiwan, which banned animal testing for cosmetics in 2013 and 2016, respectively. Now, California, is poised to do the same. Last month, California State Assembly passed bill SB 1249, which makes it illegal to sell products that have been, or include ingredients that have been, tested on animals. If signed into law by Governor Jerry Brown, the change will go into effect on Jan. 1, 2020, and would apply to all brands currently selling in California as well as any others that enter the market.
The Independent Cosmetic Manufacturers and Distributors (ICMAD) noted inconsistencies by California legislators, as the state advocates animal testing by accepting data from animal tests to determine if a chemical/ingredient should be listed on its Proposition 65 as a possible carcinogen or reproductive toxicant. So, while with this legislation, California is looking to ban animal testing, with Prop 65, it is requiring animal testing.
“Make no mistake, no one is advocating that animal testing be continued. What we do believe is that attacking our industry is the wrong approach,” concluded ICMAD. “We believe this issue is one that needs to be addressed directly by supporting the development of alternate testing models and reference data bases and not by passing laws that are purely sensational and have no real effect. ICMAD will keep working towards national uniformity on this issue and others.”
For its part, ICMAD supports the Safe Cosmetics Modernization Act (HR575), a bill introduced by Rep. Pete Sessions (R-TX). The bill would require registration of cosmetic manufacturers, reporting on cosmetic ingredients and reporting of adverse events, but includes no provisions for FDA regulation.
ICMAD isn’t alone in calling for national uniformity on cosmetic regulation. Two bills are stuck in the US Senate that would revamp the US Food and Drug Administration’s ability to regulate cosmetics. In May, 2017, Senators Dianne Feinstein (D-CA) and Susan Collins (R-ME), reintroduced the Personal Care Products Safety Act (S1113), which would require the FDA to review five cosmetic ingredients a year and would specify the first five. A year ago, Senator Orrin Hatch (R-UT) introduced S2003, a bill that NGOs call a “less ambitious alternative” to S1113.
According to Baubie, while both Senate bills would require mandatory registration of cosmetic facilities, ingredient reporting and review, only the Feinstein-Collins bill would give the FDA authority to order product recalls and the authority to annually investigate the safety of specified ingredients. The Feinstein-Collins bill also includes a “fee” provision aimed at providing the resources needed to fund the FDA’s proposed oversight.
“Any new regulations will no doubt have an industry impact,” noted Baubie. “It is not clear, however, what that impact will be, and it will remain unclear until the legislation is refined into law. While one might expect that such legislation would be opposed by the major industry players, that does not appear to be case. Several of the largest beauty and personal care product brands have confirmed their public support for such legislation.”
But whatever proposal you prefer, Arent Fox attorneys don’t expect any movement on either bill prior to the seating of the next Congress.
Importantly, neither bill was able to move forward towards passage and demonstrate significant support in Congress at this time. Thus, the next Congress will be the next opportunity to introduce such legislation and work for passage, according to Georgia C. Ravitz, partner, Arent Fox, a Washington, DC law firm which specializes in the cosmetic industry.
“We recognize that there are a number of committed members in Congress like Feinstein who are committed to increasing industry oversight and will continue to look for opportunities to move such legislation forward,” said Ravitz. “But given the anti-regulation climate in DC, we don’t believe the current Congress and Administration are sufficiently supportive of passing new regulations of this type unless a major safety incident were to occur to change the landscape for cosmetics reform. However, a new Congress will begin in January 2019 and we will look closely then to assess the possibilities and priorities that materialize.”
When Congress finally takes up the issue of cosmetic reform, it should provide a lift for testing companies—and new costs for cosmetic companies and their suppliers, as industry experts predict any new regulations will include, an annual user fee similar to what is currently in place at FDA for drugs and devices.
“Ostensibly, the user fees would be used by FDA to increase staff so the agency could expand its safety compliance programs and related enforcement and regulatory oversight,” said Naomi Halpern, counsel, Arent Fox.
Given the new regulatory requirements included in each bill, the impacts on industry would include increased ingredient and finished product testing to substantiate ingredient/product safety, according to Halpern. Cosmetic GMPs would also increase facility compliance costs, particularly for smaller facilities that don’t have the resources to devote to expanding GMP compliance, such as equipment calibration, retrofitting, increased record keeping, reporting and safety substantiation testing, she added.
Arent Fox experts note that the FDA Cosmetic Safety and Modernization Act and the Personal Care Products Safety Act are largely similar to the comprehensive cosmetic reform proposals that have been introduced in each recent Congress going back more than 10 years—but neither proposal has ever really had enough support for passage. Similar to past measures, these bills include provisions that would require: facility registration; submission of cosmetic ingredient statements; adverse event reporting; the establishment of cosmetic GMPs; record keeping; and greater focus on ingredient safety and the role of non-functional ingredients, among other things, according to James Hartten, senior public affairs and regulatory analyst, Arent Fox.
“Both bills were introduced in 2017 and referred to the Senate Health Committee. We see the prospects for passage of a broad cosmetics reform measure as very low in the current Congress, as there is very little time left in this session, and with this being an election year and the current anti-regulation climate in DC. Plus, since introduction, neither bill has been moved forward in Committee,” explained Hartten. “So really no chance of passing this session and both measures will sunset at the end of 2018. However, similar legislation will likely be introduced again in 2019 and if either the Senate of House were to flip the chances of passage of a cosmetics reform measure would certainly improve.”
According to Arent Fox, these bills increase the focus on cosmetic ingredient safety and being able to substantiate the safety of both ingredients and finished products, including having testing data to support product/ingredient safety. Were such a bill to be enacted, it is unlikely that testing companies would be adversely affected and may see greater demand for testing services.
And while the Feinstein-Collins bill would give the FDA authority to order product recalls and the authority to annually investigate the safety of specified ingredients, Arent Fox attorneys pointed out that based on their experience, companies are often willing to conduct voluntary recalls when legitimate safety issues exists, so the practical consequence of greater FDA recall authority may be not be impactful. This bill also directs FDA to identify a small number of ingredients each year that would be investigated for safety—a little like a Cosmetic Ingredient Review (CIR), which could be utilized in an advisory role should this type of review process ever to be implemented, according to Arent Fox.
Global Aspirations
While the world waits on Washington, testing companies continue to expand their testing services into new product categories and geographic regions. Dermscan offers tests on all types of population, climates and cosmetic habits. For these reasons, the company is already present in Europe (France and Poland) as well as in Thailand and Tunisia. To complete its offer, last year, Dermscan established a working relationship with Insight Research, Mauritius.
Wherever testing takes place, Sirvent pointed out that each region of the world has its own regulations and CROs should have an expert knowledge of each one in order to advise its customers about their best options.
“Thus, in the US, the conditions for evaluating the SPF are not the same as in Europe,” she said. “In France, the law ‘Jardé’ supervises clinical tests and it is important to take it into account, even in the context of a study on a cosmetic product.”
CRG is continuously building its capabilities to keep pace with the growth demands of its industry sponsors, according to Baubie. For example, the company met industry demands by building a state-of-the-art hot room for antiperspirant studies, training and certifying malodor assessors. It regularly engages specialized medical personnel as the need for dermatologists, ophthalmologists, neo-natal and pediatric professionals arise. This year is no exception. Industry innovation in hair care products is in high-demand, so this year CRG has focused on expanding its hair expertise.
“We have recently broadened our hair assessment capabilities to include certified hair graders in hair color, tone, texture, condition, flexibility, shine, and percentage gray,” explained Baubie.
“Additionally, we are expanding our expertise in dandruff grading. CRG has one of the most experienced and sought-after dandruff graders in the country and we want to continue to build on that expertise by ensuring that we have a second generation of dandruff graders who have been rigorously trained and qualified by a leading expert.”
Finally, while CRG is a Minnesota company, Baubie and her team pride themselves on being able to service client needs, wherever those needs arise.
“We have the demonstrated capability to take our “show-on-the-road,” she said. “We have successfully partnered with other entities both nationally and internationally, to ensure that our expertise extends well-beyond our Minnesota borders. Our willingness and ability to take our know-how on the road, keeps our costs highly competitive, since our overhead remains centralized, rather than exponentially multiplied across several regional sites.”
No matter where a testing company’s headquarters are located, if its clients do business in the US, any regulatory action is expected to create more demand for testing services.
A list of testing companies begins below.
Testing Service Providers
Below is a list of testing providers serving the household and personal products industry. For more information, contact the testing service provider using the information provided for each firm.
Should your company be listed here? Contact Happi to be included in our next testing services directory. Send your request to tbranna@rodmanmedia.com.
• Alliance Technologies, LLC
Monmouth Junction, NJ
Tel: (877) 962-5993
Fax: (732) 438-8265
Email: info@alliancetechgroup.com
Website: www.alliancetechgroup.com
Testing Specialties: Alliance Technologies is a full service, DEA-licensed, and FDA-registered and audited contract laboratory offering a wide range of chemical analysis and material testing services to a diverse, international client base. Our expertise and years of experience enable us to select the correct analytical approaches and generate the accurate data needed to solve your toughest problem.
• AMA Laboratories, Inc.
New City, NY
Tel: (845) 634-4330
Fax: (845) 634-5565
Email: sales@amalabs.com
Website: www.amalabs.com
Testing Specialties: AMA Laboratories conducts state-of-the-art testing in RIPT/Safety, Infrared Testing (IRPF), SPF Determinations, Claim Substantiation with Biophysical Measurements, In-Vitro Studies and unique Matched Scientific Photography (MSP) using AMA’s PhotoGrammetrix (PhGx) System. Advanced studies can enhance documented final report data with our PolyChrommetrix (PcMx) 3D modeling plus corporate and advertising video capabilities. The AMA system offers a proprietary concept of scientifically acquired, non-invasive “before and after” images which can be electronically measured. This unique system allows for matched high resolution photographs to be directly incorporated into our clients’ advertising and marketing materials for a truly sensational presentation format.
• Ametek Brookfield
Middleboro, MA
Tel: (508) 946-6200
Email: MA-MID.info@ametek.com
Website: www.brookfieldengineering.com
• Beta Analytic Inc.
Miami, FL
Tel: (305) 662-7760
Email: lab@radiocarbon.com
Website: www.radiocarbon.com
Testing Specialties: Bio-based content and natural products testing services; high-quality results in only 5-7 business days (faster service available) under standards that include ASTM D6866, CEN 16137, EN 16640, ISO 16620, ISO 19984. Results can be accessed online 24/7 including QA reports and sample photos.
• BioScreen Testing Services, Inc.
Torrance, CA
Tel: (310) 214-0043
Fax: (310) 370-3642
Email: info@bioscreen.com
Website: www.bioscreen.com
Testing Specialties: BioScreen Testing Services is a full service, GMP, human clinical, microbiology and analytical chemistry testing laboratory. BioScreen’s clinical services division conducts product safety and efficacy claim studies using a complete array of scientific instrumentation. The company also has sophisticated image capture and analysis software to provide the highest level of scientific image claim support. BioScreen’s clinical services include safety tests for irritation and sensitivity (HRIPT) on the skin and complete ophthalmologic safety tests. BioScreen specializes in customized studies to support product claims from moisturization, anti-aging, SPF and acne, to specialized scar, cellulite, esthetic and eyelash studies. Also offered are Asian subject panels and consumer use studies. BioScreen offers a complete array of analytical chemistry to serve OTC/cosmetic clients from raw material ingredients, sunscreen assay and heavy metals to photo stability, stability, and toxicological assessments. Our microbiological department conducts a compliment of specialized micro studies and ID methods plus more routine preservative testing, microbial plate count, log reduction and bacteriostasis studies.
• Bria Research Labs and Testing Services
Libertyville, IL
Tel: (847) 918-1774
Fax: (847) 816-1830
Email: arun@brialab.com
Website: www.brialab.com
• Canfield Scientific, Inc.
Parsippany, NJ
Tel: (973) 434-1200 or (800) 815-4330
Email: info@canfieldsci.com
Website: www.canfieldsci.com
• Ceutical Laboratories, Inc.
Farmers Branch, TX
Tel: (972) 241-8374
Fax: (972) 241-0619
Email: info@ceuticallabs.com
Website: www.ceuticallabs.com
• Charles River
Charleston, SC
Tel: 877.CRIVER.1 (877-274-8371)
Website: www.criver.com
• Clinical Research
Laboratories, LLC
Piscataway, NJ
Tel: (732) 981-1616
Fax: (732) 981-0520
Email: info@crlresearchlabs.com
Website: www.CRLResearchLabs.com
Testing Specialties: Clinical Research Laboratories, LLC (CRL) operates as an independent contract laboratory providing a wide range of clinical safety and efficacy testing to the cosmetic, beauty products, personal care and pharmaceutical industries. Located both in central New Jersey and Winston-Salem, North Carolina, CRL is dedicated to conducting human clinical test procedures to determine the safety and efficacy of cosmetic, personal care, and OTC drug products. The company focuses its clinical expertise in the areas of dermatology, photobiology, ophthalmology, bioinstrumentation, microbiology, cosmetology, clinical trials and associated services.
• Comprehensive Research Group, Inc.
Minneapolis, MN
Tel: (612) 781-3400
Email: tbaubie@crginc.org
Website: crginc.org
Testing Specialties: For over 20 years, CRG has provided quality data to personal and beauty care manufacturers. We provide superior subject recruitment, study execution and data collection for product development, efficacy, and claims. We have extensive expertise in hair care, skin care, anti-aging treatments, antiperspirants/deodorants, baby care, grooming and safety-in-use testing.
• Consumer Product Testing Company, Inc.
Fairfield, NJ
Tel: (973) 609-5747
Fax: (973) 253-4059
Email: sales@cptcplabs.com
Website: www.cptclabs.com
Testing Specialties: CPTC has provided a full service GMP/GLP/GCP testing laboratory for the cosmetic, personal care, medical device, pharmaceutical and specialty chemical markets. Our capabilities include services in the clinical safety and efficacy, chemistry/microbiology/stability, photobiology, in-vitro toxicological fields and consultation. Our focus is on providing “Conception to Completion” service. We are proud to announce the addition of “Gluten Free” testing, FDA Import Detention testing and new “Bundle” pricing on Predictive Patch, Preservative and Ocular Irritation studies.
• Dermatest Medical Research
Münster, Germany
Tel: (49) 251-481637-0
Fax: (49) 251-481637-66
Email: info@dermatest.de
Website: www.dermatest.de
• Dermscan Group
Lyon, France
Tel: +33(0) 472 823 656
Email: commercial@dermscan.com
Website: www.dermscan.com
Testing Specialties: Dermscan is the reference in clinical trials of cosmetics and pharmaceutical products since 1990 and also realize pre-clinical in vitro tests in GLP certified laboratory. ISO 9001:2015 certified, its facilities in France, Poland, Tunisia, Thailand, Mauritius and international partnerships, provide them multi ethnic panels for standard or complex multiparametric studies.
• EAG Laboratories
San Diego, CA
Tel: (877) 709-9526
Website: www.eag.com
• Essex Testing Clinic, Inc.
Verona, NJ
Tel: 973-857-9541
Email: info@essextesting.com,
Don@essextesting.com
Website: www.essextesting.com
Testing Specialties: Human clinical evaluations performed for safety, efficacy and claims support of cosmetics, raw materials, toiletries, personal care, household, and dermal OTCs. With two facilities and a diverse subject base we specialize in full range of tests, including but not limited to RIPT, SPF, In-Use, Anti-Aging, Patch, photosensitivity, Long Lasting, Waterproof, Comedogenicity, eye products, clinical and perception studies. Protocols developed for specific needs.
• Evalulab Inc.
Montreal, QC, Canada
Tel: (514) 343-0001
Website: www.evalulab.com
Sales: ekulig@evalulab.com
Testing Specialties: Evalulab is a contract research organization offering standard and customized testing services to evaluate safety, performance and consumer preference of skin care, cosmetics, personal care products, medical devices and raw materials. The laboratory is certified ISO 9001:2015 and provides clinical studies that comply to international GCP, FDA and Health Canada.
• Genemarkers, LLC
Kalamazoo, MI
Tel: (844) 220-6231
Email: info@genemarkersllc.com
Website: www.genemarkersllc.com
Testing Specialties: Genemarkers is a contract testing lab that specializes in genomics. Core services include genotyping, gene expression and microRNA profiling. The technology is used to identify the biological mechanisms of action of raw materials and finished products. Testing is used to optimize product development and to validate efficacy.
• Harrison Research
Laboratories, Inc.
Union, NJ
Tel: (908) 688-7600
Fax: (908) 688-7601
Email: client@hrlabs.us.com
Website: www.hrlabs.us.com
• Hill Top Research Incorporated
St. Petersburg, FL
Tel: (727) 344-7602
Email: info@hill-top.com
Website: www.hill-top.com
• Idea Tests Group
Martillac, France
Tel: +33 556 64 82 33
Contact: Frederic Nunzi
Email: f.nunzi@groupeideatests.com
Website: www.groupeideatests.com
Testing Specialties: Idea Tests Group specializes in the evaluation of cosmetic and personal care products for both ingredients and final formulations. Idea Tests Group provides a holistic approach and a wide range of services: in vitro OECD toxicology, efficacy, and microbiology tests, clinical studies to assess tolerance and efficacy, regulatory expertise to support your specific situation, in vitro and in vivo suncare product testing. With seven evaluation centers Idea Tests Group provides its services to customers all over the world.
• Impact Analytical
Midland, MI
Tel: (855) 427-6583
Email: info@impactanalytical.com
Website: www.ImpactAnalytical.com
• International Research Services, Inc.
Port Chester, NY
Tel: (914) 937-9483, (800) 548-0158
Email: info@irsi.org
Website: www.irsi.org
• InVitro International
Placentia, CA 92870
Tel: (800) 246-8487, (949) 851-8356
Fax: (949) 851-4985
Email: invitro@invitrointl.com
Website: www.invitrointl.com
Testing Specialties: Least cost, fastest, validated as accurate—Ocular Irritection and Dermal Irritection. Final reports in as few as 36 hours; accuracy proven as high as 90+% vs animal results. More than a quarter century of satisfied customers with a published peer reviewed OECD study result available. GHS accepted Corrositex: least cost, fastest, and most accurate global leader in non-animal corrosion testing; results possible in 24 hours or less.
• MB Research Laboratories
Spinnerstown, PA
Tel: 215-536-4110
Fax: 215-536-1816
Email: customerservice@mbresearch.com
Website: www.mbresearch.com
Testing Specialties: MB Research Labs has been dedicated to conducting cosmetic and personal care safety assessments for over 40 years. Specialized services include, but are not limited to, in vitro dermal and eye irritation, dermal sensitization, and phototoxicity. An award-winning recognized leader in the development and use of in vitro/alternative toxicology.
• McCrone Group
Westmont, IL
Tel: (630) 887-7100
Website: www.mccroneassociates.com
• Micro Quality Labs Inc. (MQL Inc.)
Burbank, CA
Tel: (818) 845-0070
Fax: (818) 845-0030
Website: www.microqualitylabs.com
Testing Services: Micro Quality Labs is an ISO 17025 certified lab, established in 2003 and offers a comprehensive range of contract testing services to the cosmetic, nutritional, pharmaceutical, OTC and household products industries. Services include Analytical, Stability Testing (ICH guidelines), Heavy Metals, Microbiology, Method Development and Validation, Environmental Testing, Water Testing, Pesticides/USP, 1,4 Dioxane, Amino Acid Testing and Phthalates.
• Princeton Consumer Research
Princeton, NJ
Tel: (609) 455-1112
Fax: (609) 580-1393
Email: janetervooren@princetonconsumer.com
Website: princetonconsumer.com
Testing Specialties: Princeton Consumer Research is a global consumer product testing company specializing in safety, efficacy and acceptability studies. PCR focuses on the cosmetic, skin, beauty, personal care, and baby care industries. Their global headquarters in Princeton, NJ and their site in St. Petersburg, FL have the largest environmentally controlled rooms and hot rooms in the US. PCR also has 2 additional sites in the UK, and a site in Winnipeg, Canada. Plans include expansion to Asia and Brazil in 2018-2019.
• Product Integrity Laboratory LLC
Landing, NJ
Tel: (973) 770-7707
Fax: (973) 770-7711
Email: info@prodinteglab.com
Website: www.prodinteglab.com
Technical Services: Product development and formulation, quality control and quality assurance, regulatory compliance, production troubleshooting.
• Q Laboratories, Inc.
Cincinnati, OH
Tel: (513) 471-1300
Fax: (513) 471-5600
Email: mgoins@qlaboratories.com
Website: www.qlaboratories.com
Testing Specialties: An FDA registered, cGMP/GLP compliant, ISO/IEC 17025 accredited analytical chemistry and microbiology laboratory. Services include: Method development and validation, microbial detection and enumeration, microbial identification, active ingredient analysis, raw material qualification, antimicrobial efficacy analysis and stability studies.
• SGS
Fairfield, NJ
Tel: (973) 575-5252
Fax: (973) 575-7175
Email: uscts.inquiries@sgs.com
Website: www.sgs.com/cosmetics
Testing Specialties: It’s crucial for cosmetics and personal care products to be effective, stable and safe. SGS’ global quality assurance and performance services can help you ensure the quality, safety, efficacy and regulatory compliance of your products. Laboratory and field operations offer tailor-made solutions to meet your chemical, physical, microbiological and claim support needs.
• Skinobs
France
Tel: +33(0)630089098
Email: contact@skinobs.com
Website: www.skinobs.com
Testing Specialties: Analytical tests, toxicology regulatory, tolerance, use tests, ex vivo, in vitro and in vivo efficacy, sensory analysis for skin care, body care, solar, capillary and makeup.
• Solar Light Company, Inc.
Glenside, PA
Tel: (215) 517-8700
Fax: (215) 517-8747
Email: info@solarlight.com
Website: www.SolarLight.com