Nava Dayan, Dr. Nava Dayan LLC11.02.16
Cells emerged on earth at least 3.5 billion years ago, but it took humanity quite some time to discover and then explore these magical structural units. Cells were discovered in 1665 by Robert Hooke, an English natural philosopher, architect and mathematician. But it wasn’t until 1839 that scientists acknowledged all organisms are composed of one or more cells. At this time, the cell was defined as the fundamental unit of structure and function in living organisms. Although cell biology research has advanced greatly in recent decades, there are many barriers that science has yet to overcome in order to leap to the next level in understanding cell biology. Scientists have been successful in isolation, assembly, manipulation and acquiring limited control over cell populations; but so far this is done at the expense of understanding integration and communication between cell populations.
Adding to the complexity, livelihood and cellular homeostasis is often confused and misunderstood. There is a natural law of biological systems striving to homeostasis; which I define as a “state of temporary biological satisfaction under specific conditions.” Note that homeostasis is not necessarily a healthy condition but a preferred state dictated by specific circumstance; therefore a disease is homeostasis that has been created to achieve best conditions at a time point. Many confuse homeostasis with stagnation. When a living organism lacks activity it is not alive. Inactivity is death. Homeostasis, therefore, is a state of energy consumption and constant change associated effort and is powered by continual adaptations at the cellular, organ and body level. Current research is limited in two key areas: commune and environmental interactions and kinetics. Cells interact and respond to other cells, energies, nutrients, hormones, cytokines, biota and many other factors, some yet to be discovered. Isolation of cells, as done for research, manipulating and exposing cells to an artificial environment, may not at all reflect their normal behavior. Analyzing cell function at selected time intervals may result in additional inaccuracies in their lifecycle. An important change that may take milliseconds and is not detected can generate misconception and wrongful conclusions. While we can learn from techniques currently available, one should bear in mind the great limitations of present research platforms.
Maintenance of cell populations descended from a single cell (termed “cell lines”) became a laboratory technique only in the middle of the 20th century. The ability to maintain cells, allow proliferation and differentiation out of the organ and body environment, became an important expertise that is used across a variety of research areas. Cells grown in culture require specific conditions to survive, grow, function and proliferate. Some of these conditions include:
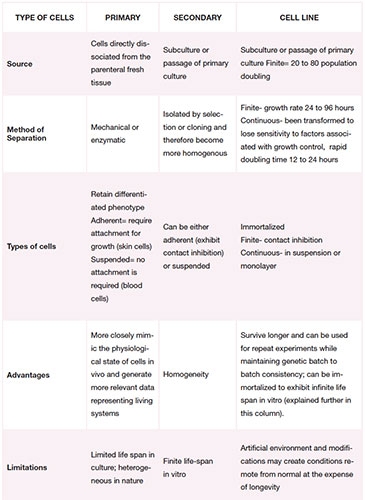
Cell cultures can be classified into three isolation and development categories as outlined in the table:
An immortalized cell line is composed of cells collected from a multicellular organism which normally when isolated would not proliferate indefinitely. In order to allow long-life span proliferation rate to eliminate the need of re-harvesting from an organ and maintain genetic identity throughout a variety of experiments, these cells undergo mutation. This mutation eliminates the normal cell aging process (senescence) and allows them to proliferate beyond normal. In many ways this genetic modification turns them close to cancer cells in phenotype and the cells can therefore grow for prolonged periods in vitro.
It’s important to note that such mutations can occur naturally and such cells may be isolated from cancer tissues; or they can be caused intentionally when they are induced for experimental purposes. If genetically identical cell populations are required for comparison experiments, the immortalized cells can be cloned giving rise to clonal population.
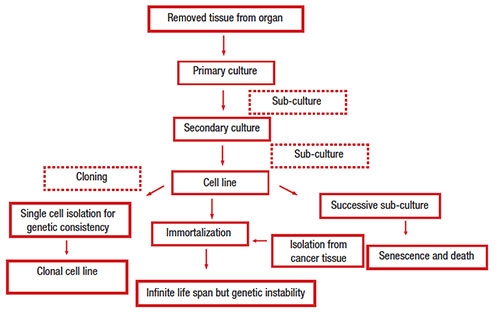
The scheme below illustrates isolation processes of cell lines for in vitro culture:
As mentioned, all in vitro and ex vivo models present artifacts because they are detached from the body and placed in an unnatural environment. In addition, most of these systems require maintaining sterility, which deletes any activity associated with environmental insult such as the microbiome or pollution.
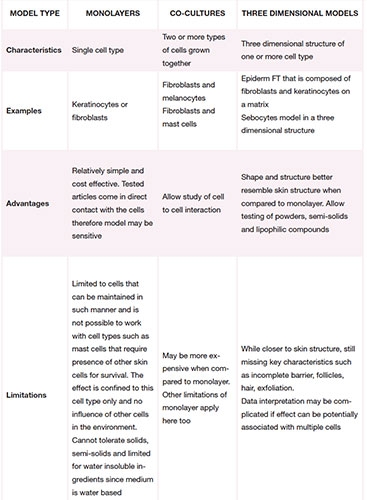
Still, there are choices available to consider. The table left summarizes the three most common systems, their characteristics, advantages and limitations.
Another model that is often used is skin explants; that is, utilizing fresh excised skin for studying the effect of interaction with ingredients or formulations. While using fresh human skin may seem the ultimate choice for research because it is a real tissue, this option presents a few key limitations:
Until recently, it had been nearly impossible to work with isolated sebocytes, due to their functional and structural complexity and activity. Recent advancements gave rise to a variety of interesting models to study the effect of ingredients and formulations on sebum secretion and other functions in sebocytes models. Cultured human sebocytes preserve important sebocytic characteristics, although they undergo an incomplete terminal differentiation in vitro.
Over the years, modifications of the technique have improved the culture of human sebocytes in vitro, but the primary cultured sebocytes can still be maintained for no more than six passages.
The immortalized human sebaceous gland cell lines SZ95, SEB-1 and Seb-E6E7 have been developed in recent years, which allow the extraction of a large number of sebocytes from the same donor culture. The study of cultured human sebocytes in vitro has become a useful tool in examining sebaceous gland activity and regulation, and understanding the pathophysiological mechanisms and treatment of acne and other sebaceous gland related diseases.
Although such a model can be instrumental for initial screening, it may pose some limitations because of the mere isolation, changes associated with immortalization, loss of its location in the gland, variations in lipid synthesis, difficulties to separate sebum and stratum corneum produced lipids and alike.
A more advanced model can utilize an isolated human sebaceous gland. Culture of the entire sebaceous gland offers several advantages in comparison to sebocyte monolayer culture.
For example, as the three-dimensional architecture is maintained, proliferation and lipid synthesis can be simultaneously examined in the basal and differentiated cell layers, respectively. Another advantage over an animal model that may be associated with hair, such system permits delivery of potential sebum suppressive compounds to the gland so biological activity can be evaluated with no obstacles.
In summary in vitro and ex vivo cellular biological systems are available and used extensively in basic and industrial research. It is considered a cost effective and relatively simple approach to understand biological cascades.
Yet, the following considerations should be exercised in any such experiment:
Nava Dayan
Owner
Dr. Nava Dayan LLC
Nava Dayan Ph.D. is the owner of Dr. Nava Dayan L.L.C, a skin science and research consultancy and serving the pharmaceutical, cosmetic, and personal care industries. She has 25 years of experience in the skin care segment, and more than 150 publication credits. Tel: 201-206-7341; E-mail: nava.dayan@verizon.net
Adding to the complexity, livelihood and cellular homeostasis is often confused and misunderstood. There is a natural law of biological systems striving to homeostasis; which I define as a “state of temporary biological satisfaction under specific conditions.” Note that homeostasis is not necessarily a healthy condition but a preferred state dictated by specific circumstance; therefore a disease is homeostasis that has been created to achieve best conditions at a time point. Many confuse homeostasis with stagnation. When a living organism lacks activity it is not alive. Inactivity is death. Homeostasis, therefore, is a state of energy consumption and constant change associated effort and is powered by continual adaptations at the cellular, organ and body level. Current research is limited in two key areas: commune and environmental interactions and kinetics. Cells interact and respond to other cells, energies, nutrients, hormones, cytokines, biota and many other factors, some yet to be discovered. Isolation of cells, as done for research, manipulating and exposing cells to an artificial environment, may not at all reflect their normal behavior. Analyzing cell function at selected time intervals may result in additional inaccuracies in their lifecycle. An important change that may take milliseconds and is not detected can generate misconception and wrongful conclusions. While we can learn from techniques currently available, one should bear in mind the great limitations of present research platforms.

- Substrate or liquid such as flask or scaffold material; chemically modified plastic or coated with extra cellular matrix proteins;
- Culture media that contain nutrients;
- An appropriate environment such as gas CO2, temperature (37°C), humidity; and
- Sterility that may be achieved by utilizing aseptic technique, antibiotics and antimycotic agents.
Cell cultures can be classified into three isolation and development categories as outlined in the table:
An immortalized cell line is composed of cells collected from a multicellular organism which normally when isolated would not proliferate indefinitely. In order to allow long-life span proliferation rate to eliminate the need of re-harvesting from an organ and maintain genetic identity throughout a variety of experiments, these cells undergo mutation. This mutation eliminates the normal cell aging process (senescence) and allows them to proliferate beyond normal. In many ways this genetic modification turns them close to cancer cells in phenotype and the cells can therefore grow for prolonged periods in vitro.
It’s important to note that such mutations can occur naturally and such cells may be isolated from cancer tissues; or they can be caused intentionally when they are induced for experimental purposes. If genetically identical cell populations are required for comparison experiments, the immortalized cells can be cloned giving rise to clonal population.
The scheme below illustrates isolation processes of cell lines for in vitro culture:
As mentioned, all in vitro and ex vivo models present artifacts because they are detached from the body and placed in an unnatural environment. In addition, most of these systems require maintaining sterility, which deletes any activity associated with environmental insult such as the microbiome or pollution.

Another model that is often used is skin explants; that is, utilizing fresh excised skin for studying the effect of interaction with ingredients or formulations. While using fresh human skin may seem the ultimate choice for research because it is a real tissue, this option presents a few key limitations:
- Population diversity requires large sample size for statistical significance;
- The tissue is detached from the body, therefore is not connected to the blood circulation, no exfoliation occurs; and
- Regulatory obstacles, difficult to obtain and need to work with it when fresh.
Until recently, it had been nearly impossible to work with isolated sebocytes, due to their functional and structural complexity and activity. Recent advancements gave rise to a variety of interesting models to study the effect of ingredients and formulations on sebum secretion and other functions in sebocytes models. Cultured human sebocytes preserve important sebocytic characteristics, although they undergo an incomplete terminal differentiation in vitro.
Over the years, modifications of the technique have improved the culture of human sebocytes in vitro, but the primary cultured sebocytes can still be maintained for no more than six passages.
The immortalized human sebaceous gland cell lines SZ95, SEB-1 and Seb-E6E7 have been developed in recent years, which allow the extraction of a large number of sebocytes from the same donor culture. The study of cultured human sebocytes in vitro has become a useful tool in examining sebaceous gland activity and regulation, and understanding the pathophysiological mechanisms and treatment of acne and other sebaceous gland related diseases.
Although such a model can be instrumental for initial screening, it may pose some limitations because of the mere isolation, changes associated with immortalization, loss of its location in the gland, variations in lipid synthesis, difficulties to separate sebum and stratum corneum produced lipids and alike.
A more advanced model can utilize an isolated human sebaceous gland. Culture of the entire sebaceous gland offers several advantages in comparison to sebocyte monolayer culture.

In summary in vitro and ex vivo cellular biological systems are available and used extensively in basic and industrial research. It is considered a cost effective and relatively simple approach to understand biological cascades.
Yet, the following considerations should be exercised in any such experiment:
- Careful selection of model;
- Culture conditions;
- Detailed protocol;
- Tested time point;
- Biological paths;
- Analytical methods;
- Controls—positive, negative, know marketable actives; and
- Genetics and genetics consistency across batches.
Nava Dayan
Owner
Dr. Nava Dayan LLC
Nava Dayan Ph.D. is the owner of Dr. Nava Dayan L.L.C, a skin science and research consultancy and serving the pharmaceutical, cosmetic, and personal care industries. She has 25 years of experience in the skin care segment, and more than 150 publication credits. Tel: 201-206-7341; E-mail: nava.dayan@verizon.net