01.02.19
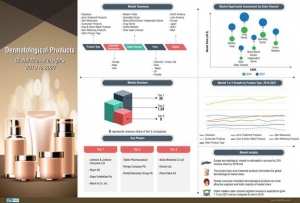
The US Food & Drug Administration (FDA) issued a Request for Information (RFI) and established a docket to obtain data, information and comments that will assist the agency in assessing the safety and effectiveness of OTC food handler antiseptic drug products; i.e., antiseptic hand washes or rubs intended for use in food handling settings. The FDA has tentatively concluded that food handler antiseptics may differ from antiseptic products addressed in other rulemakings. This tentative determination is based on the current categorization of other antiseptic products and considers factors that may include specific microorganisms of concern in food handling environments, as well as the safety of repeated-exposure use patterns. It also considers issues raised by the public health consequences of foodborne illness, differences in frequency and type of use, and contamination of the hands by dirt, grease and other oils. There has been support from industry and interested parties for an OTC food handler antiseptic category. Some information and data have already been submitted to the FDA in support of establishing such a category. The FDA said more information is needed to assist the agency in evaluating the safety and effectiveness criteria appropriate for food handler antiseptics.
Through this RFI, the agency is asking manufacturers of food handler antiseptics and other interested parties to submit safety and effectiveness data on OTC food handler antiseptics marketed for commercial or regulated environments where growth, harvest, production, manufacturing, processing, packaging, transportation, storage, preparation, service or consumption of food occurs. The FDA is also seeking comments and requesting data on definitions, eligibility, current conditions of use of food handler antiseptics, safety and effectiveness criteria, as well as test methods to demonstrate the safety and effectiveness of food handler antiseptics.
This information and data will inform the agency’s ongoing review of OTC antiseptic drug products.
Through this RFI, the agency is asking manufacturers of food handler antiseptics and other interested parties to submit safety and effectiveness data on OTC food handler antiseptics marketed for commercial or regulated environments where growth, harvest, production, manufacturing, processing, packaging, transportation, storage, preparation, service or consumption of food occurs. The FDA is also seeking comments and requesting data on definitions, eligibility, current conditions of use of food handler antiseptics, safety and effectiveness criteria, as well as test methods to demonstrate the safety and effectiveness of food handler antiseptics.
This information and data will inform the agency’s ongoing review of OTC antiseptic drug products.